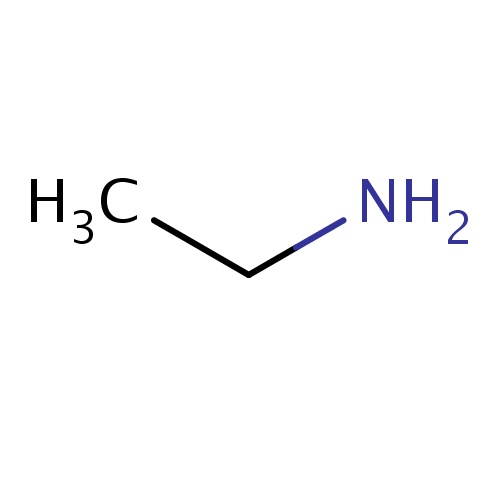
Common name
ethanamine
IUPAC name
ethanamine
SMILES
CCN
Common name
ethanamine
IUPAC name
ethanamine
SMILES
CCN
INCHI
InChI=1S/C2H7N/c1-2-3/h2-3H2,1H3
FORMULA
C2H7N

Common name
ethanamine
IUPAC name
ethanamine
Molecular weight
45.084
clogP
-0.554
clogS
-0.083
Frequency
0.0677
HBond Acceptor
0
HBond Donor
2
Total PolarSurface Area
26.02
Number of Rings
0
Rotatable Bond
0
| Drug ID | Common name | Structure CAS | Compound class | Therapeutic area |
|---|---|---|---|---|
| FDBD01101 | Lapatinib |
|
Antineoplastic Agents; Protein Kinase Inhibitors; Antineoplastic and Immunomodulating Agents; Cytochrome P-450 CYP2C8 Inhibitors; Cytochrome P-450 CYP2C19 Inducers; Cytochrome P-450 CYP2C8 Inducers; CYP3A4 Inhibitors; Combined Inhibitors of CYP3A4 and P-glycoprotein; | Indicated in combination with capecitabine for the treatment of patients with advanced or metastatic breast cancer whose tumors overexpress the human epidermal receptor type 2 (HER2) protein and who have received prior therapy including an anthracycline, a taxane, and trastuzuma. |
| FDBD01103 | Sitagliptin |
|
Hypoglycemic Agents; Dipeptidyl-Peptidase IV Inhibitors; Drugs Used in Diabetes; Alimentary Tract and Metabolism; Blood Glucose Lowering Drugs, Excl. Insulins; Cytochrome P-450 CYP2C8 Inhibitors; Cytochrome P-450 CYP2C8 Inducers; Incretins; CYP3A4 Inhibitors; | For use as an adjunct to diet and exercise to improve glycemic control in patients with type 2 diabetes mellitus. Also for use in patients with type 2 diabetes mellitus to improve glycemic control in combination with metformin or a PPARγ agonist (e.g., thiazolidinediones) when the single agent alone, with diet and exercise, does not provide adequate glycemic control. |
| FDBD01109 | Sunitinib |

|
Antineoplastic Agents; Immunosuppressive Agents; Angiogenesis Inhibitors; Protein Kinase Inhibitors; Antineoplastic and Immunomodulating Agents; CYP3A4 Inhibitors; | For the treatment of advanced renal cell carcinoma as well as the treatment of gastrointestinal stromal tumor after disease progression on or intolerance to imatinib mesylate. |
| FDBD01111 | Arformoterol |

|
Adrenergic beta-2 Receptor Agonists; Bronchodilator Agents; Cytochrome P-450 CYP2C9 Inhibitors; Cytochrome P-450 CYP2C9 Inducers; Cytochrome P-450 CYP2C19 Inducers; CYP2A6 Inhibitors; CYP2A6 Inhibitors (strong); CYP2A6 Inhibitors (moderate); CYP2A6 Inducers; CYP2A6 Inducers (strong); CYP2D6 Inducers; CYP2D6 Inducers (strong); Beta2 Agonists; | A bronchodilator used for the long term, symptomatic treatment of reversible bronchoconstriction in patients with chronic obstructive pulmonary disease (COPD), including chronic bronchitis and emphysema. |
| FDBD01116 | Fenoterol |

|
Sympathomimetics; Adrenergic beta-2 Receptor Agonists; Bronchodilator Agents; Tocolytic Agents; Respiratory System; Drugs for Obstructive Airway Diseases; Genito Urinary System and Sex Hormones; Selective Beta-2-Adrenoreceptor Agonists; Adrenergics, Inhalants; Adrenergics for Systemic Use; Sympathomimetics, Labour Repressants; Beta2 Agonists; | Fenoterol is used for the treatment of asthma. |
| FDBD01120 | Bevantolol |
|
Adrenergic beta-1 Receptor Antagonists; Adrenergic alpha-1 Receptor Antagonists; Cardiovascular System; Beta Blocking Agents, Selective; Beta Blocking Agents; Beta Blocking Agents, Selective, and Thiazides; Beta Blocking Agents and Thiazides; | For the treatment of angina pectoris and hypertension. |
| FDBD01127 | Fosamprenavir |
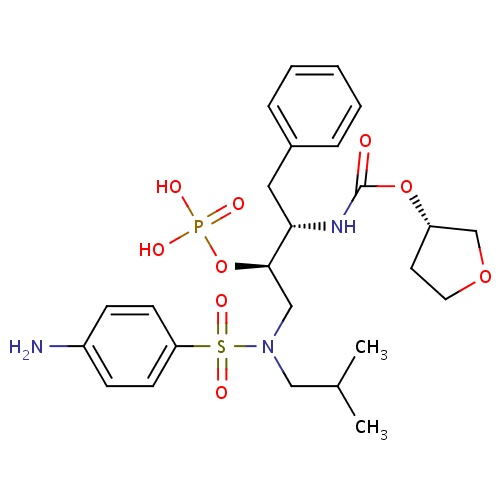
|
Anti-HIV Agents; Protease Inhibitors; HIV Protease Inhibitors; Prodrugs; Antiinfectives for Systemic Use; Direct Acting Antivirals; Antivirals for Systemic Use; CYP3A4 Inhibitors; | Indicated in combination with other antiretroviral agents for the treatment of human immunodeficiency virus (HIV-1) infection, as well as postexposure prophylaxis of HIV infection in individuals who have had occupational or nonoccupational exposure to potentially infectious body fluids of a person known to be infected with HIV when that exposure represents a substantial risk for HIV transmission. The use of fosamprenavir is pending revision due to a potential association between the drug and myocardial infarction and dyslipidemia in HIV infected adults. |
| FDBD01144 | Cilazapril |
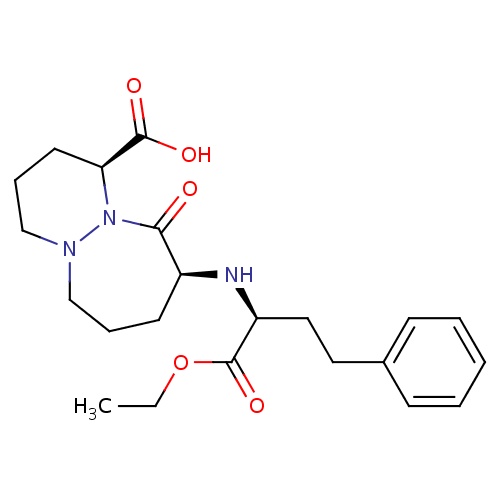
|
Angiotensin-Converting Enzyme Inhibitors; Antihypertensive Agents; Cardiovascular System; Agents Acting on the Renin-Angiotensin System; ACE Inhibitors, Plain; ACE Inhibitors and Diuretics; | Cilazapril is an ACE inhibtor class drug used in the treatment of hypertension and heart failure. |
| FDBD01148 | Spirapril |
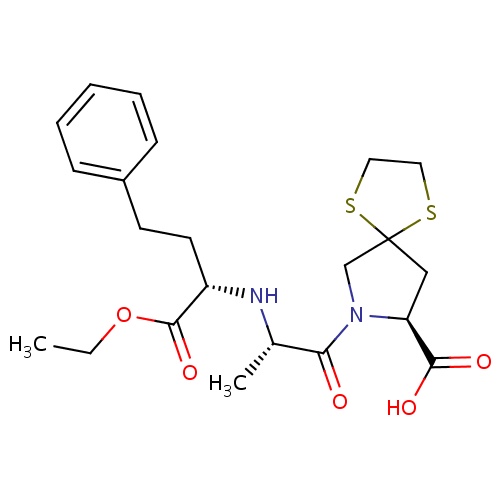
|
Cardiovascular System; Agents Acting on the Renin-Angiotensin System; ACE Inhibitors, Plain; | Spirapril is an ACE inhibitor class drug used to treat hypertension. |
| FDBD01204 | Aprindine |
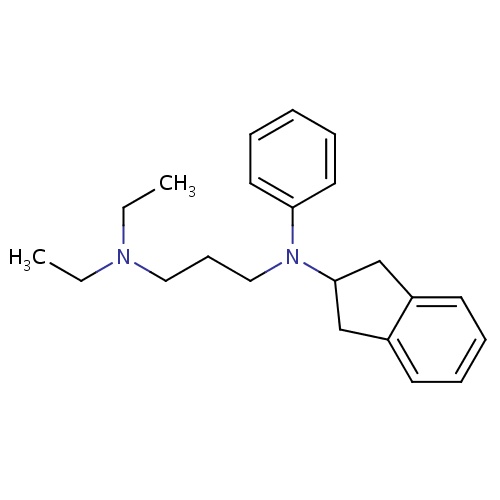
|
Anti-Arrhythmia Agents; Voltage-Gated Sodium Channel Blockers; Cardiovascular System; Antiarrhythmics, Class I and Iii; Cardiac Therapy; Antiarrythmics, Class I and Iii; Antiarrhythmics, Class Ib; CYP2D6 Inducers; CYP2D6 Inducers (strong); |
197 ,
20
| FRAGNAME | PDBID | SIMILIRITY | XSCORE | SMILE | HAC |
|---|---|---|---|---|---|
| 1a99_ligand_1_2.mol2 | 1a99 | 1 | -5.92 | CC[NH3+] | 3 |
| 1pot_ligand_1_5.mol2 | 1pot | 1 | -5.90 | C([NH3+])C | 3 |
| 1i7m_ligand_1_2.mol2 | 1i7m | 1 | -5.88 | CC[NH3+] | 3 |
| 3a5y_ligand_1_0.mol2 | 3a5y | 1 | -5.86 | [NH3+]CC | 3 |
| 2zxg_ligand_frag_1.mol2 | 2zxg | 1 | -5.85 | C([NH3+])C | 3 |
| 3ebh_ligand_1_0.mol2 | 3ebh | 1 | -5.84 | [NH3+]CC | 3 |
| 4q4i_ligand_1_0.mol2 | 4q4i | 1 | -5.83 | [NH3+]CC | 3 |
1149 ,
115

