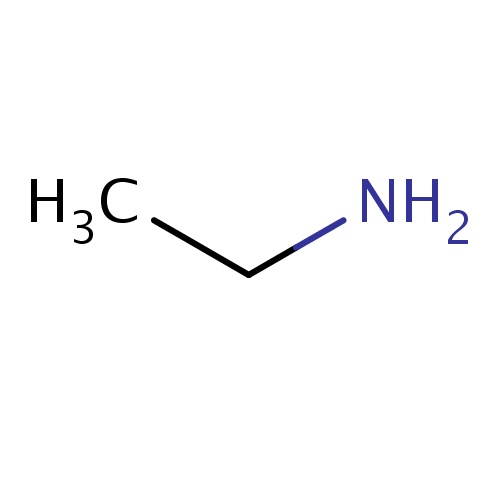
Common name
ethanamine
IUPAC name
ethanamine
SMILES
CCN
Common name
ethanamine
IUPAC name
ethanamine
SMILES
CCN
INCHI
InChI=1S/C2H7N/c1-2-3/h2-3H2,1H3
FORMULA
C2H7N

Common name
ethanamine
IUPAC name
ethanamine
Molecular weight
45.084
clogP
-0.554
clogS
-0.083
Frequency
0.0677
HBond Acceptor
0
HBond Donor
2
Total PolarSurface Area
26.02
Number of Rings
0
Rotatable Bond
0
| Drug ID | Common name | Structure CAS | Compound class | Therapeutic area |
|---|---|---|---|---|
| FDBD00753 | Benzylpenicilloyl Polylysine |
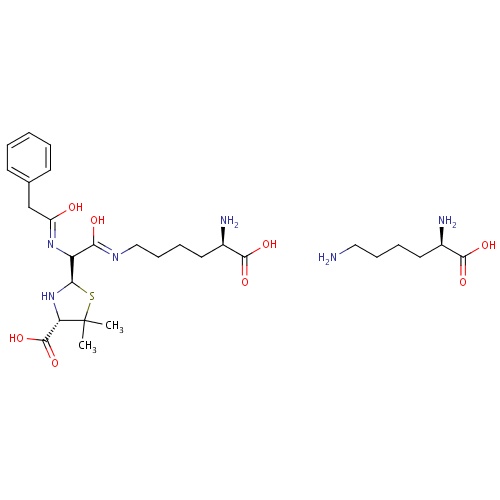
|
Acetamides; | For use as a adjunct in assessing the risk of administering penicillin (benzylpenicillin or penicillin G). |
| FDBD00782 | Phenoxybenzamine |
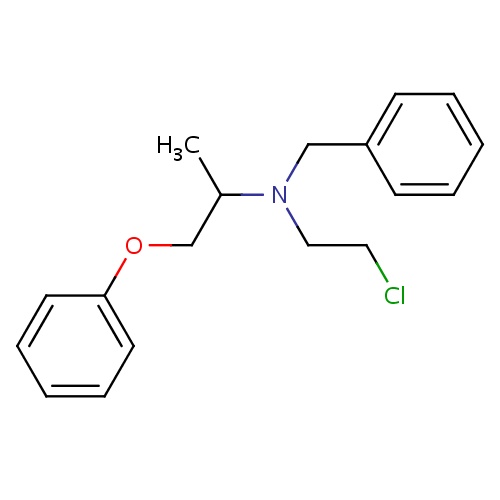
|
Antihypertensive Agents; Vasodilator Agents; Adrenergic alpha-1 Receptor Antagonists; Adrenergic alpha-Antagonists; Cardiovascular System; Peripheral Vasodilators; CYP3A4 Inhibitors; | For the treatment of phaeochromocytoma (malignant), benign prostatic hypertrophy and malignant essential hypertension. |
| FDBD00792 | Diethylpropion |

|
Appetite Depressants; Stimulants; Alimentary Tract and Metabolism; Centrally Acting Antiobesity Products; Antiobesity Preparations, Excl. Diet Products; | Used in the management of exogenous obesity as a short-term adjunct (a few weeks) in a regimen of weight reduction based on caloric restriction. |
| FDBD00793 | Salmeterol |
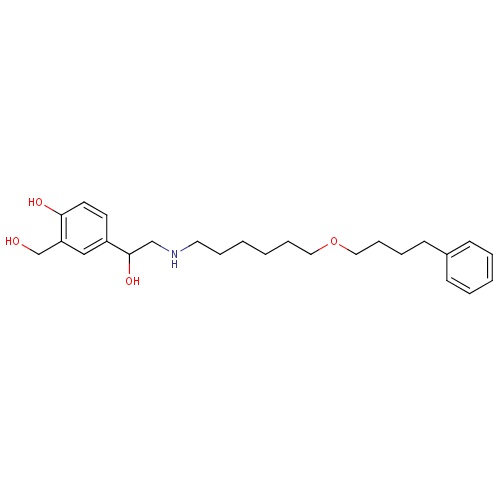
|
Sympathomimetics; Adrenergic beta-2 Receptor Agonists; Bronchodilator Agents; Respiratory System; Drugs for Obstructive Airway Diseases; Selective Beta-2-Adrenoreceptor Agonists; Adrenergics, Inhalants; Cytochrome P-450 CYP2C8 Inhibitors; Cytochrome P-450 CYP2C8 Inducers; CYP3A4 Inhibitors; Beta2 Agonists; | For the treatment of asthma and chronic obstructive pulmonary disease (COPD). |
| FDBD00810 | Netilmicin |
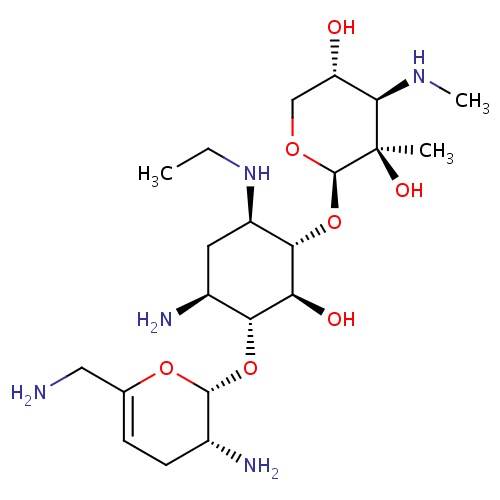
|
Anti-Bacterial Agents; Protein Synthesis Inhibitors; Aminoglycosides; Antibiotics; Ophthalmologicals; Sensory Organs; Antibacterials for Systemic Use; Antiinfectives for Systemic Use; Antiinfectives; Aminoglycoside Antibacterials; | For the treatment of bacteremia, septicaemia, respiratory tract infections, skin and soft-tissue infection, burns, wounds, and peri-operative infections caused by susceptible strains. |
| FDBD00828 | Edetic Acid |

|
Anticoagulants; Chelating Agents; Food Additives; Cytochrome P-450 CYP1A2 Inhibitors; Cytochrome P-450 CYP1A2 Inducers; Calcium Chelating Agents; | For the reduction of blood levels and depot stores of lead in lead poisoning (acute and chronic) and lead encephalopathy, in both pediatric populations and adults. |
| FDBD00837 | Formoterol |

|
Sympathomimetics; Adrenergic beta-2 Receptor Agonists; Bronchodilator Agents; Respiratory System; Drugs for Obstructive Airway Diseases; Selective Beta-2-Adrenoreceptor Agonists; Adrenergics, Inhalants; Cytochrome P-450 CYP2C9 Inhibitors; Cytochrome P-450 CYP2C9 Inducers; Cytochrome P-450 CYP2C19 Inducers; CYP2A6 Inhibitors; CYP2A6 Inhibitors (strong); CYP2A6 Inhibitors (moderate); CYP2A6 Inducers; CYP2A6 Inducers (strong); CYP2D6 Inducers; CYP2D6 Inducers (strong); Beta2 Agonists; | For use as long-term maintenance treatment of asthma in patients 6 years of age and older with reversible obstructive airways disease, including patients with symptoms of nocturnal asthma, who are using optimal corticosteroid treatment and experiencing regular or frequent breakthrough symptoms requiring use of a short-acting bronchodilator. Not indicated for asthma that can be successfully managed with occasional use of an inhaled, short-acting beta2-adrenergic agonist. Also used for the prevention of exercise-induced bronchospasm, as well as long-term treatment of bronchospasm associated with COPD. |
| FDBD00842 | Dopamine |
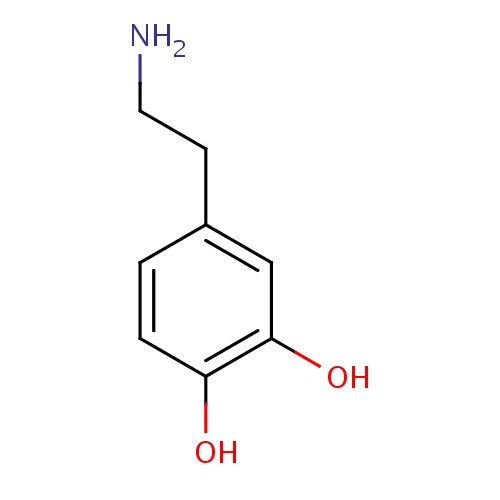
|
Dopamine Agents; Sympathomimetics; Cardiotonic Agents; Cardiovascular System; Cardiac Therapy; Adrenergic and Dopaminergic Agents; Cardiac Stimulants Excl. Cardiac Glycosides; Cytochrome P-450 CYP1A2 Inhibitors; Cytochrome P-450 CYP2C9 Inhibitors; Cytochrome P-450 CYP1A2 Inducers; Cytochrome P-450 CYP2C9 Inducers; Cytochrome P-450 CYP2C19 Inducers; CYP2D6 Inducers; CYP2D6 Inducers (strong); | For the correction of hemodynamic imbalances present in the shock syndrome due to myocardial infarction, trauma, endotoxic septicemia, open-heart surgery, renal failure, and chronic cardiac decompensation as in congestive failure. |
| FDBD00866 | Cinacalcet |
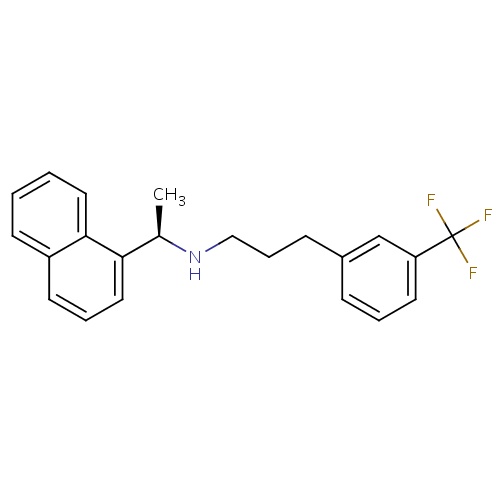
|
Systemic Hormonal Preparations, Excl. Sex Hormones and Insulins; Anti-Parathyroid Agents; Calcium Homeostasis; Cytochrome P-450 CYP1A2 Inhibitors; Cytochrome P-450 CYP1A2 Inducers; Calcimimetic Agents; CYP2D6 Inducers; CYP2D6 Inducers (strong); CYP3A4 Inhibitors; | For the treatment of secondary hyperparathyroidism in patients with Chronic Kidney Disease who are on hemodialysis or peritoneal dialysis. Also for the treatment of hypercalcemia in patients with parathyroid carcinoma. |
| FDBD00888 | Procainamide |
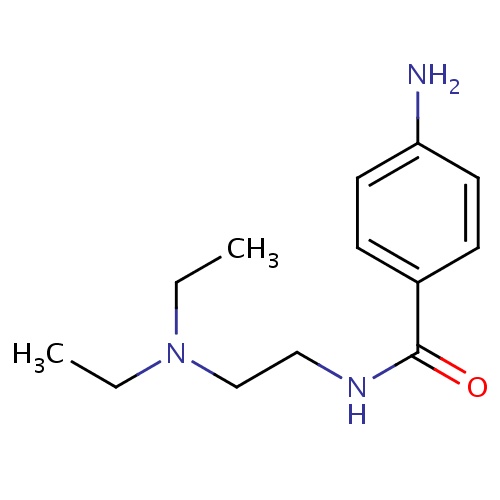
|
Anti-Arrhythmia Agents; Voltage-Gated Sodium Channel Blockers; Cardiovascular System; Antiarrhythmics, Class I and Iii; Cardiac Therapy; Antiarrythmics, Class I and Iii; Antiarrhythmics, Class Ia; CYP2D6 Inducers; CYP2D6 Inducers (strong); | For the treatment of life-threatening ventricular arrhythmias. |
197 ,
20
| FRAGNAME | PDBID | SIMILIRITY | XSCORE | SMILE | HAC |
|---|---|---|---|---|---|
| 1a99_ligand_1_2.mol2 | 1a99 | 1 | -5.92 | CC[NH3+] | 3 |
| 1pot_ligand_1_5.mol2 | 1pot | 1 | -5.90 | C([NH3+])C | 3 |
| 1i7m_ligand_1_2.mol2 | 1i7m | 1 | -5.88 | CC[NH3+] | 3 |
| 3a5y_ligand_1_0.mol2 | 3a5y | 1 | -5.86 | [NH3+]CC | 3 |
| 2zxg_ligand_frag_1.mol2 | 2zxg | 1 | -5.85 | C([NH3+])C | 3 |
| 3ebh_ligand_1_0.mol2 | 3ebh | 1 | -5.84 | [NH3+]CC | 3 |
| 4q4i_ligand_1_0.mol2 | 4q4i | 1 | -5.83 | [NH3+]CC | 3 |
1149 ,
115

