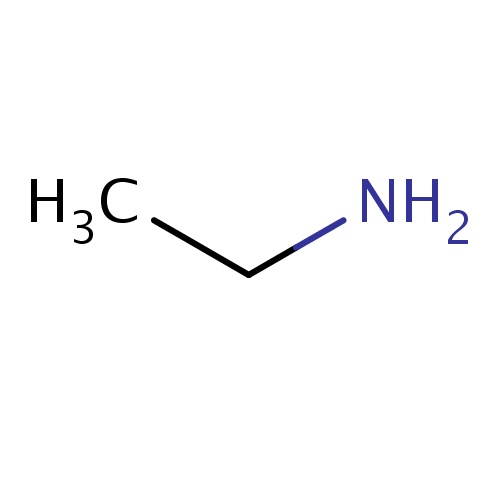
Common name
ethanamine
IUPAC name
ethanamine
SMILES
CCN
Common name
ethanamine
IUPAC name
ethanamine
SMILES
CCN
INCHI
InChI=1S/C2H7N/c1-2-3/h2-3H2,1H3
FORMULA
C2H7N

Common name
ethanamine
IUPAC name
ethanamine
Molecular weight
45.084
clogP
-0.554
clogS
-0.083
Frequency
0.0677
HBond Acceptor
0
HBond Donor
2
Total PolarSurface Area
26.02
Number of Rings
0
Rotatable Bond
0
| Drug ID | Common name | Structure CAS | Compound class | Therapeutic area |
|---|---|---|---|---|
| FDBD00889 | Tolterodine |
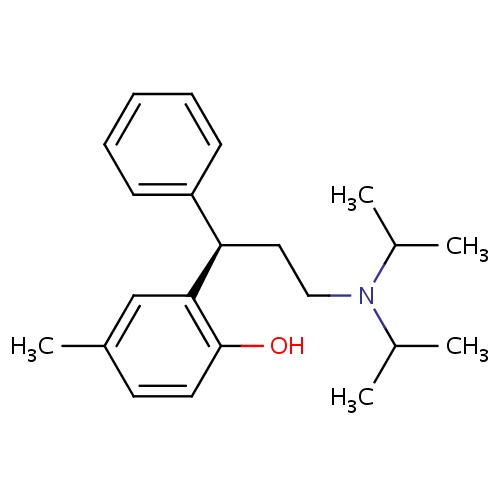
|
Muscarinic Antagonists; Anti-Incontinence Agents; Antispasmodics; Muscle Relaxants, Genitourinary; Genito Urinary System and Sex Hormones; Drugs for Urinary Frequency and Incontinence; Cytochrome P-450 CYP2C9 Inhibitors; Urological Agents; Cytochrome P-450 CYP2C9 Inducers; Cytochrome P-450 CYP2C19 Inducers; CYP2D6 Inducers; CYP2D6 Inducers (strong); CYP3A4 Inhibitors; | For the treatment of overactive bladder (with symptoms of urinary frequency, urgency, or urge incontinence). |
| FDBD00907 | Tocainide |
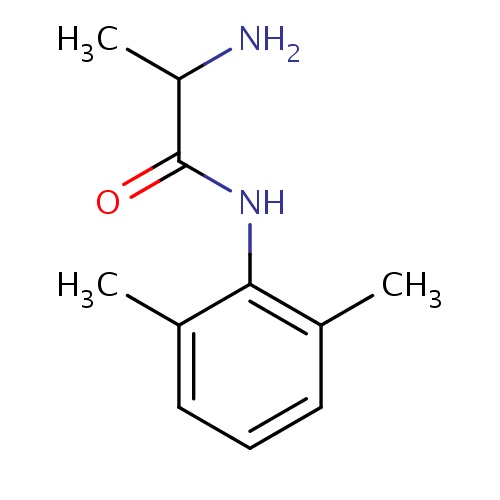
|
Anti-Arrhythmia Agents; Voltage-Gated Sodium Channel Blockers; Sodium Channel Blockers; Cardiovascular System; Antiarrhythmics, Class I and Iii; Cardiac Therapy; Antiarrythmics, Class I and Iii; Antiarrhythmics, Class Ib; Cytochrome P-450 CYP1A2 Inhibitors; Cytochrome P-450 CYP1A2 Inducers; | For the treatment of documented ventricular arrhythmias, such as sustained ventricular tachycardia, that, in the judgment of the physician, are life-threatening. |
| FDBD00913 | Oxybutynin |
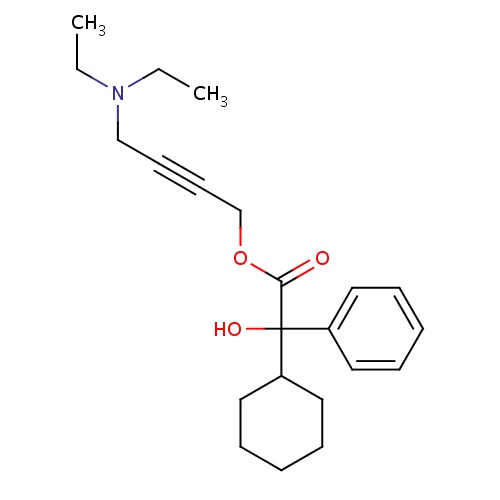
|
Muscarinic Antagonists; Parasympatholytics; Cholinergic Antagonists; Antispasmodics; Muscle Relaxants, Genitourinary; Genito Urinary System and Sex Hormones; Drugs for Urinary Frequency and Incontinence; Urological Agents; Cytochrome P-450 CYP2C8 Inhibitors; Cytochrome P-450 CYP2C8 Inducers; CYP2D6 Inducers; CYP2D6 Inducers (strong); CYP3A4 Inhibitors; | For the treatment of overactive bladder. |
| FDBD00930 | Vigabatrin |
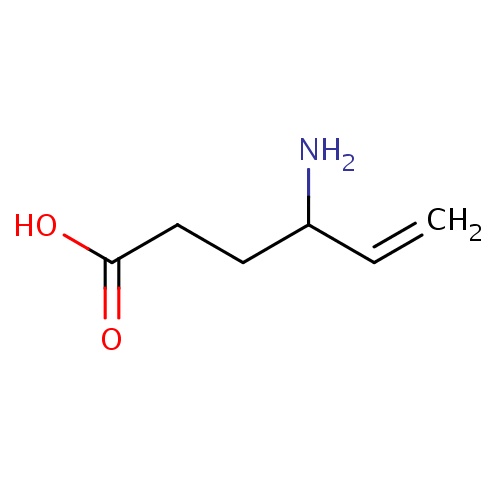
|
Enzyme Inhibitors; Anticonvulsants; GABA Agents; Nervous System; Antiepileptics; Fatty Acid Derivatives; | For use as an adjunct in treatment resistant epilepsy, refractory complex partial seizures, and secondary generalized seizures. It is also used as monotherapy in infantile spasms in West syndrome. |
| FDBD00937 | Primaquine |
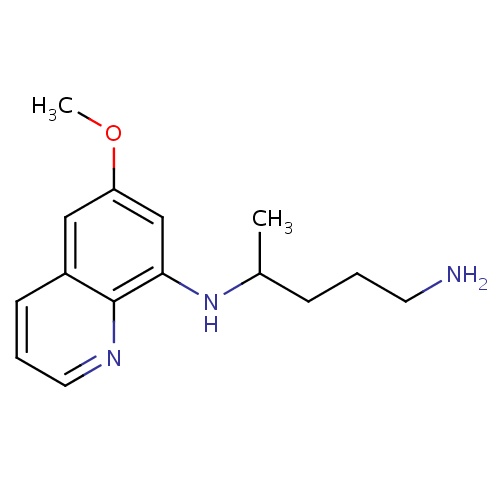
|
Antimalarials; Antiprotozoal Agents; Antiparasitic Products, Insecticides and Repellents; Aminoquinolines; Cytochrome P-450 CYP1A2 Inhibitors; Cytochrome P-450 CYP1A2 Inducers; CYP2D6 Inducers; CYP2D6 Inducers (strong); CYP3A4 Inhibitors; | For the treatment of malaria. |
| FDBD00952 | Arbutamine |
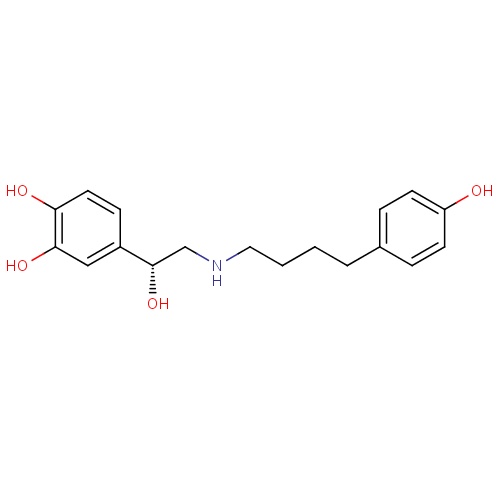
|
Cardiovascular System; Cardiac Therapy; Adrenergic and Dopaminergic Agents; Cardiac Stimulants Excl. Cardiac Glycosides; Beta2 Agonists; | Used to elicit acute cardiovascular responses (cardiac stumulant), similar to those produced by exercise, in order to aid in diagnosing the presence or absence of coronary artery disease (CAD) in patients who cannot exercise adequately. |
| FDBD00953 | Quinacrine |
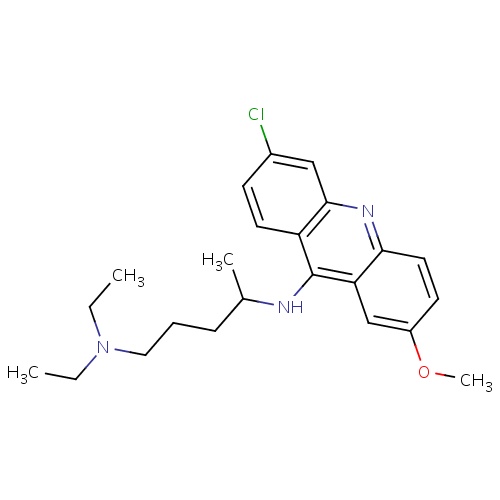
|
Antineoplastic Agents; Enzyme Inhibitors; Antimalarials; Antiprotozoal Agents; Antinematodal Agents; Anthelmintics; Anticestodal Agents; Antiparasitic Products, Insecticides and Repellents; Agents Against Protozoal Diseases; CYP3A4 Inhibitors; | For the treatment of giardiasis and cutaneous leishmaniasis and the management of malignant effusions. |
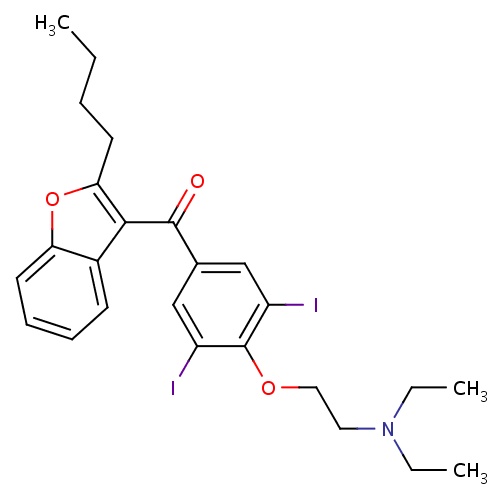
| FDBD00967 | Amiodarone |
|
Enzyme Inhibitors; Anti-Arrhythmia Agents; Potassium Channel Blockers; Vasodilator Agents; Sodium Channel Blockers; Cardiovascular System; Antiarrhythmics, Class III; Antiarrhythmics, Class I and Iii; Cardiac Therapy; Antiarrythmics, Class I and Iii; Cytochrome P-450 CYP1A2 Inhibitors; Cytochrome P-450 CYP2C9 Inhibitors; Cytochrome P-450 CYP1A2 Inducers; Cytochrome P-450 CYP2C8 Inhibitors; Cytochrome P-450 CYP3A Inhibitors; Cytochrome P-450 CYP2C9 Inducers; Cytochrome P-450 CYP2C19 Inducers; Cytochrome P-450 CYP2C8 Inducers; CYP2A6 Inhibitors; CYP2A6 Inhibitors (strong); CYP2A6 Inhibitors (moderate); CYP2A6 Inducers; CYP2A6 Inducers (strong); CYP2D6 Inducers; CYP2D6 Inducers (strong); CYP3A4 Inhibitors; Combined Inhibitors of CYP3A4 and P-glycoprotein; | Intravenously, for initiation of treatment and prophylaxis of frequently recurring ventricular fibrillation and hemodynamically unstable ventricular tachycardia in patients refractory to other therapy. Orally, for the treatment of life-threatening recurrent ventricular arrhythmias such as recurrent ventricular fibrillation and recurrent hemodynamically unstable ventricular tachycardia. |
| FDBD00971 | Ambenonium |

|
Anti-Arrhythmia Agents; Cholinesterase Inhibitors; Parasympathomimetics; Antimyasthenic Agents; Nervous System; Anticholinesterases; | Ambenonium is used to treat muscle weakness due to muscle disease (myasthenia gravis). |
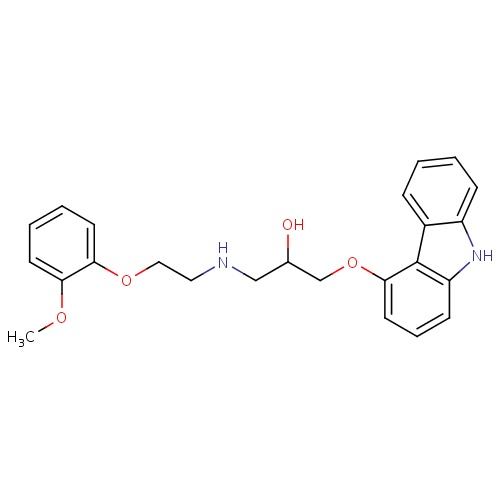
| FDBD00984 | Carvedilol |
|
Antihypertensive Agents; Vasodilator Agents; Adrenergic alpha-1 Receptor Antagonists; Adrenergic beta-Antagonists; Cardiovascular System; Beta Blocking Agents; Alpha and Beta Blocking Agents; Cytochrome P-450 CYP1A2 Inhibitors; Cytochrome P-450 CYP2C9 Inhibitors; Cytochrome P-450 CYP1A2 Inducers; Cytochrome P-450 CYP2C9 Inducers; CYP2D6 Inducers; CYP2D6 Inducers (strong); CYP2E1 Inhibitors; CYP2E1 Inducers; CYP2E1 Inducers (strong); CYP3A4 Inhibitors; | For the treatment of mild or moderate (NYHA class II or III) heart failure of ischemic or cardiomyopathic origin. |
197 ,
20
| FRAGNAME | PDBID | SIMILIRITY | XSCORE | SMILE | HAC |
|---|---|---|---|---|---|
| 1a99_ligand_1_2.mol2 | 1a99 | 1 | -5.92 | CC[NH3+] | 3 |
| 1pot_ligand_1_5.mol2 | 1pot | 1 | -5.90 | C([NH3+])C | 3 |
| 1i7m_ligand_1_2.mol2 | 1i7m | 1 | -5.88 | CC[NH3+] | 3 |
| 3a5y_ligand_1_0.mol2 | 3a5y | 1 | -5.86 | [NH3+]CC | 3 |
| 2zxg_ligand_frag_1.mol2 | 2zxg | 1 | -5.85 | C([NH3+])C | 3 |
| 3ebh_ligand_1_0.mol2 | 3ebh | 1 | -5.84 | [NH3+]CC | 3 |
| 4q4i_ligand_1_0.mol2 | 4q4i | 1 | -5.83 | [NH3+]CC | 3 |
1149 ,
115

