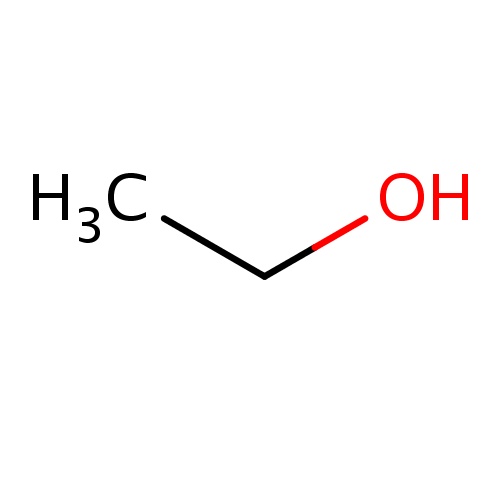
Common name
ethanol
IUPAC name
ethanol
SMILES
CCO
Common name
ethanol
IUPAC name
ethanol
SMILES
CCO
INCHI
InChI=1S/C2H6O/c1-2-3/h3H,2H2,1H3
FORMULA
C2H6O

Common name
ethanol
IUPAC name
ethanol
Molecular weight
46.068
clogP
-0.315
clogS
0.129
Frequency
0.1474
HBond Acceptor
1
HBond Donor
1
Total PolarSurface Area
20.23
Number of Rings
0
Rotatable Bond
0
| Drug ID | Common name | Structure CAS | Compound class | Therapeutic area |
|---|---|---|---|---|
| FDBD00424 | Hydroxyzine |
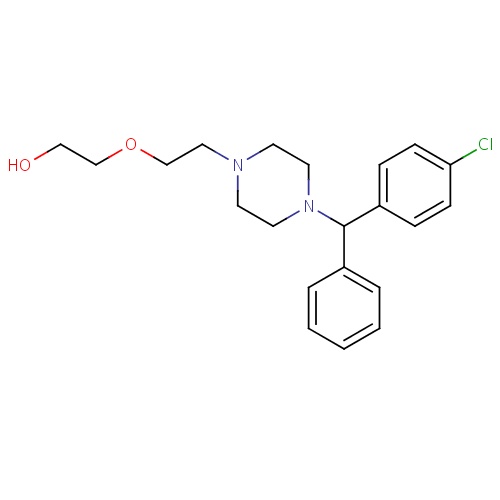
|
Hypnotics and Sedatives; Antipruritics; Histamine H1 Antagonists; Nervous System; Anxiolytics; Psycholeptics; Diphenylmethane Derivatives; CYP2D6 Inducers; CYP2D6 Inducers (strong); | For symptomatic relief of anxiety and tension associated with psychoneurosis and as an adjunct in organic disease states in which anxiety is manifested. Useful in the management of pruritus due to allergic conditions such as chronic urticaria. |
| FDBD00426 | Bosentan |
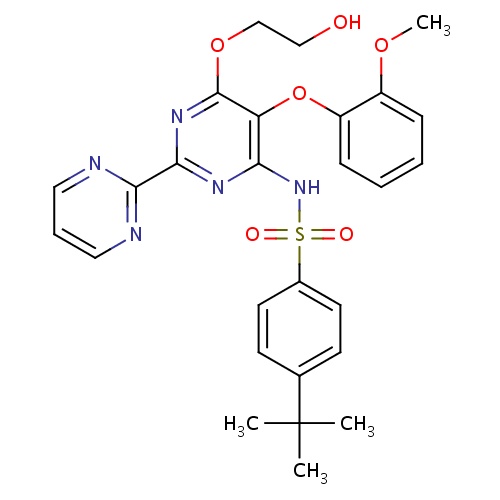
|
Antihypertensive Agents; Cardiovascular System; Antihypertensives for Pulmonary Arterial Hypertension; Cytochrome P-450 CYP2C9 Inhibitors; Cytochrome P-450 CYP2C9 Inducers; CYP3A4 Inhibitors; BSEP/ABCB11 Inhibitors; Endothelin Receptor Antagonists; | Used in the treatment of pulmonary arterial hypertension (PAH), to improve exercise ability and to decrease the rate of clinical worsening (in patients with WHO Class III or IV symptoms). |
| FDBD00438 | Propranolol |
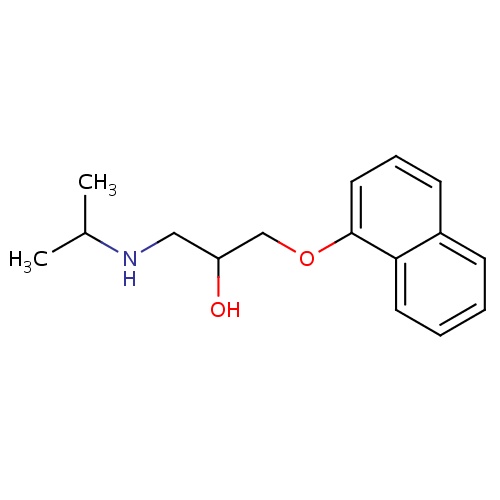
|
Antihypertensive Agents; Anti-Arrhythmia Agents; Vasodilator Agents; Adrenergic beta-Antagonists; Cardiovascular System; Beta Blocking Agents; Beta Blocking Agents and Thiazides; Beta Blocking Agents, Non-Selective; Beta Blocking Agents, Non-Selective, and Thiazides; Cytochrome P-450 CYP1A2 Inhibitors; Cytochrome P-450 CYP1A2 Inducers; Cytochrome P-450 CYP2C19 Inducers; CYP2D6 Inducers; CYP2D6 Inducers (strong); CYP3A4 Inhibitors; | For the prophylaxis of migraine. |
| FDBD00448 | L-Carnitine |
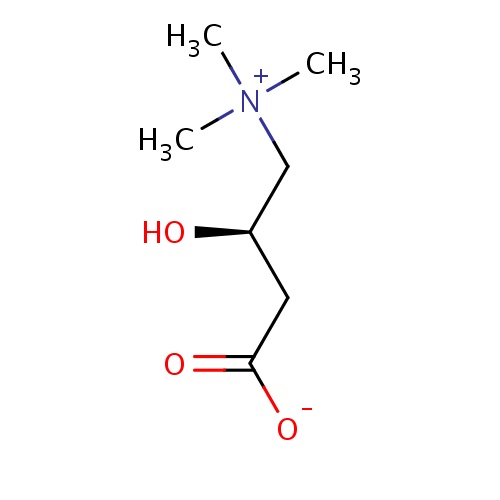
|
Vitamin B Complex; Dietary Supplements; Alimentary Tract and Metabolism; Amino Acids and Derivatives; | For treatment of primary systemic carnitine deficiency, a genetic impairment of normal biosynthesis or utilization of levocarnitine from dietary sources, or for the treatment of secondary carnitine deficiency resulting from an inborn error of metabolism such as glutaric aciduria II, methyl malonic aciduria, propionic acidemia, and medium chain fatty acylCoA dehydrogenase deficiency. Used therapeutically to stimulate gastric and pancreatic secretions and in the treatment of hyperlipoproteinemias. Parenteral levocarnitine is indicated for the prevention and treatment of carnitine deficiency in patients with end-stage renal disease. |
| FDBD00462 | Gadoteridol |
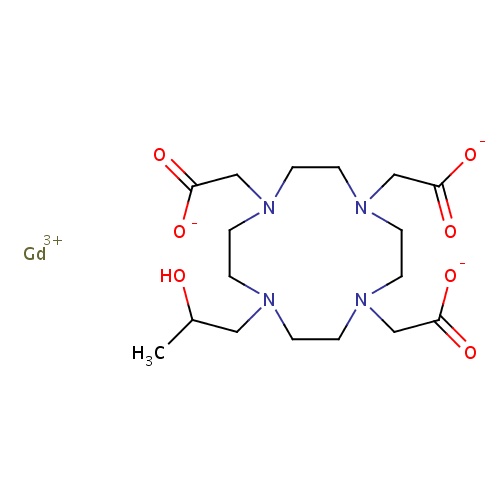
|
Contrast Media; Paramagnetic Contrast Media; Magnetic Resonance Imaging Contrast Media; | Gadoteridol is an MRI contrast agent used for contrast enhancement of the brain, spine and surrounding tissues resulting in improved visualization (compared with unenhanced MRI) of lesions with abnormal vascularity or those thought to cause a disruption of the normal blood brain barrier. Gadoteridol can also be used for whole body contrast enhanced MRI including the head, neck, liver, breast, musculoskeletal system and soft tissue pathologies. |
| FDBD00463 | Labetalol |
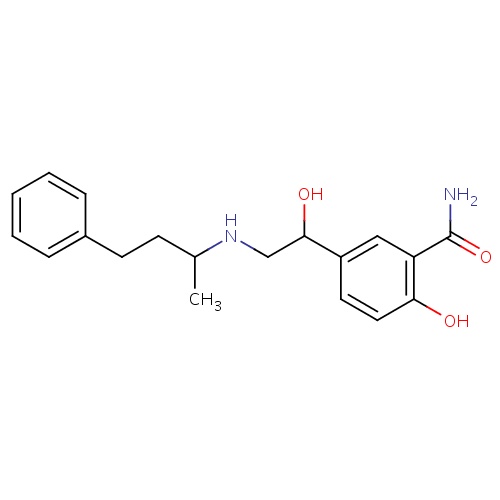
|
Antihypertensive Agents; Sympathomimetics; Adrenergic beta-Antagonists; Cardiovascular System; Beta Blocking Agents; Beta Blocking Agents and Thiazides; Alpha and Beta Blocking Agents; Alpha and Beta Blocking Agents and Thiazides; CYP2D6 Inducers; CYP2D6 Inducers (strong); | For the management of hypertension (alone or in combination with other classes of antihypertensive agents), as well as chronic stable angina pectoris and sympathetic overactivity syndrome associated with severe tetanus. Labetalol is used parenterally for immediate reduction in blood pressure in severe hypertension or in hypertensive crises when considered an emergency, for the control of blood pressure in patients with pheochromocytoma and pregnant women with preeclampsia, and to produce controlled hypotension during anesthesia to reduce bleeding resulting from surgical procedures. |
| FDBD00469 | Cisapride |
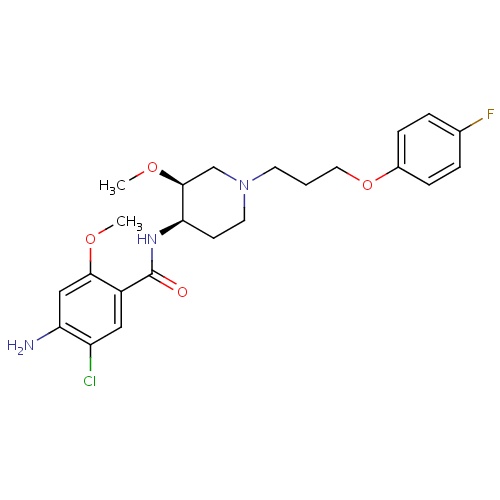
|
Gastrointestinal Agents; Anti-Ulcer Agents; Serotonin Receptor Agonists; Prokinetic Agents; Alimentary Tract and Metabolism; Drugs for Functional Gastrointestinal Disorders; Propulsives; Cytochrome P-450 CYP1A2 Inhibitors; Cytochrome P-450 CYP2C9 Inhibitors; Cytochrome P-450 CYP1A2 Inducers; Cytochrome P-450 CYP2C8 Inhibitors; Cytochrome P-450 CYP2C9 Inducers; Cytochrome P-450 CYP2C19 Inducers; Cytochrome P-450 CYP2C8 Inducers; Cytochrome P-450 CYP2B6 Inducers; Cytochrome P-450 CYP2B6 Inhibitors; CYP2A6 Inhibitors; CYP2A6 Inhibitors (strong); CYP2A6 Inhibitors (moderate); CYP2A6 Inducers; CYP2A6 Inducers (strong); CYP2B6 Inhibitors (strong); CYP2D6 Inducers; CYP2D6 Inducers (strong); CYP3A4 Inhibitors; | For the symptomatic treatment of adult patients with nocturnal heartburn due to gastroesophageal reflux disease. |
| FDBD00472 | Nafcillin |
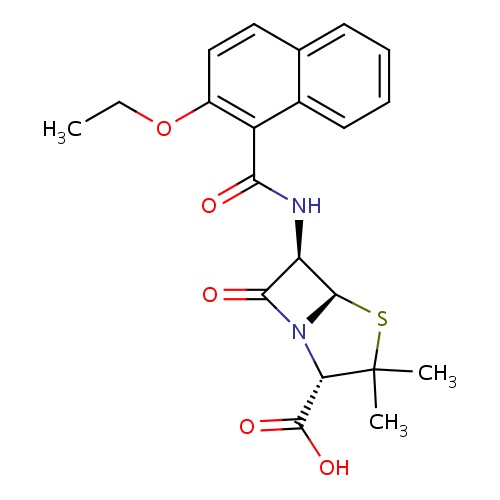
|
Anti-Bacterial Agents; Penicillins; Antibacterials for Systemic Use; Antiinfectives for Systemic Use; Beta-Lactamase Resistant Penicillins; Beta-Lactam Antibacterials, Penicillins; Cytochrome P-450 CYP1A2 Inhibitors; Cytochrome P-450 CYP1A2 Inducers; CYP3A4 Inhibitors; | For the treatment of infections caused by penicillinase-producing staphylococci which have demonstrated susceptibility to the drugs. |
| FDBD00477 | Bisoprolol |
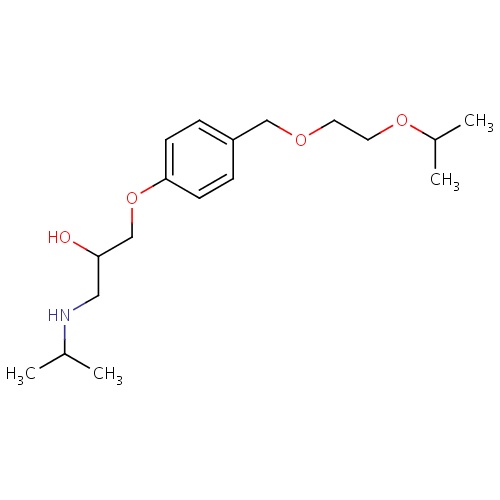
|
Antihypertensive Agents; Adrenergic beta-1 Receptor Antagonists; Sympatholytics; Cardiovascular System; Agents Acting on the Renin-Angiotensin System; Beta Blocking Agents, Selective; Beta Blocking Agents; Beta Blocking Agents, Selective, and Thiazides; Beta Blocking Agents and Thiazides; CYP2D6 Inducers; CYP2D6 Inducers (strong); CYP3A4 Inhibitors; | For management of heart failure, angina pectoris, and mild to moderate hypertension and for secondary prevention of myocardial infarction (MI). |
| FDBD00481 | Candoxatril |
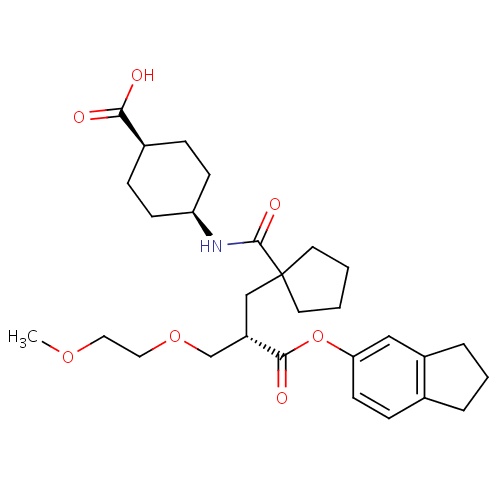
|
Prodrugs; | For treatment of hypertension, improve exercise capacity in patients with CHF receiving angiotensin converting enzyme inhibition. |
429 ,
43
| FRAGNAME | PDBID | SIMILIRITY | XSCORE | SMILE | HAC |
|---|---|---|---|---|---|
| 2reg_ligand_1_0.mol2 | 2reg | 1 | -5.87 | C(O)C | 3 |
| 3c2u_ligand_1_4.mol2 | 3c2u | 1 | -5.81 | CCO | 3 |
| 3aje_ligand_frag_1.mol2 | 3aje | 1 | -5.79 | C(O)C | 3 |
| 4bt4_ligand_frag_1.mol2 | 4bt4 | 1 | -5.74 | C(O)C | 3 |
| 4bt5_ligand_frag_1.mol2 | 4bt5 | 1 | -5.74 | C(O)C | 3 |
| 4hwp_ligand_frag_0.mol2 | 4hwp | 1 | -5.74 | CCO | 3 |
| 3c2u_ligand_1_3.mol2 | 3c2u | 1 | -5.72 | CCO | 3 |
| 4hwr_ligand_frag_2.mol2 | 4hwr | 1 | -5.72 | C(O)C | 3 |
| 4hwo_ligand_frag_2.mol2 | 4hwo | 1 | -5.71 | C(O)C | 3 |
2270 ,
228

