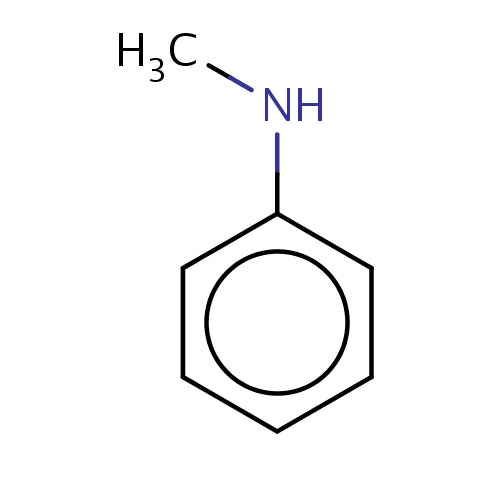
Common name
N-methylaniline
IUPAC name
N-methylaniline
SMILES
CNc1ccccc1
Common name
N-methylaniline
IUPAC name
N-methylaniline
SMILES
CNc1ccccc1
INCHI
InChI=1S/C7H9N/c1-8-7-5-3-2-4-6-7/h2-6,8H,1H3
FORMULA
C7H9N

Common name
N-methylaniline
IUPAC name
N-methylaniline
Molecular weight
107.153
clogP
1.572
clogS
-2.011
Frequency
0.0076
HBond Acceptor
0
HBond Donor
1
Total PolarSurface Area
12.03
Number of Rings
1
Rotatable Bond
1
| Drug ID | Common name | Structure CAS | Compound class | Therapeutic area |
|---|---|---|---|---|
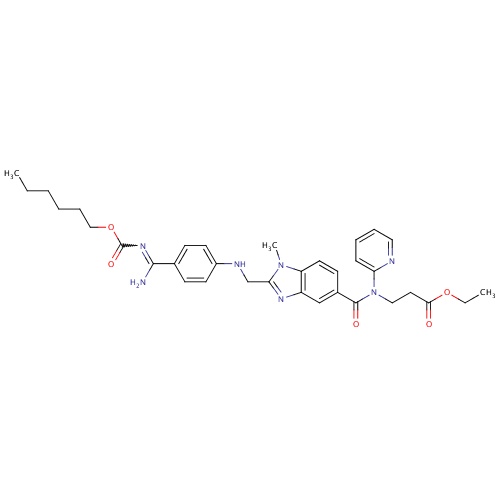
| FDBD01435 | Dabigatran etexilate |
|
Antithrombins; Direct Thrombin Inhibitors; Antithrombotic Agents; Blood and Blood Forming Organs; | Dabigatran is indicated for the prevention of venous thromboembolic events in patients who have undergone elective hip or knee replacement surgery (based on RE-NOVATE, RE-MODEL, and RE-MOBILIZE trials). In 2010, it was approved in the US and Canada for prevention of stroke and systemic embolism in patients with atrial fibrillation (approval based on the RE-LY trial). Contraindications: severe renal impairment (CrCL . |
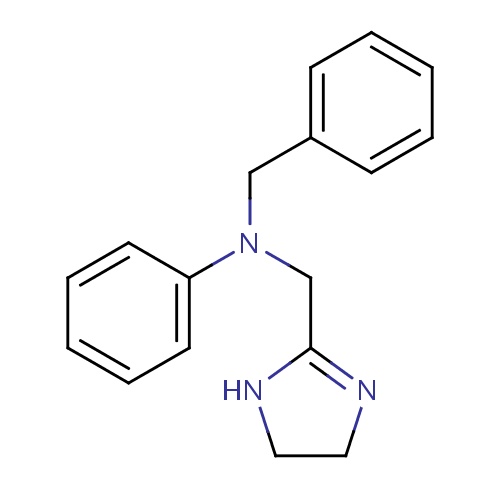
| FDBD01517 | Antazoline |
|
Anti-Allergic Agents; Histamine H1 Antagonists; Histamine Antagonists; Respiratory System; Nasal Preparations; Antihistamines for Systemic Use; Antiallergic Agents, Excl. Corticosteroids; | Used to relieve nasal congestion and in eye drops, usually in combination with naphazoline, to relieve the symptoms of allergic conjunctivitis. |
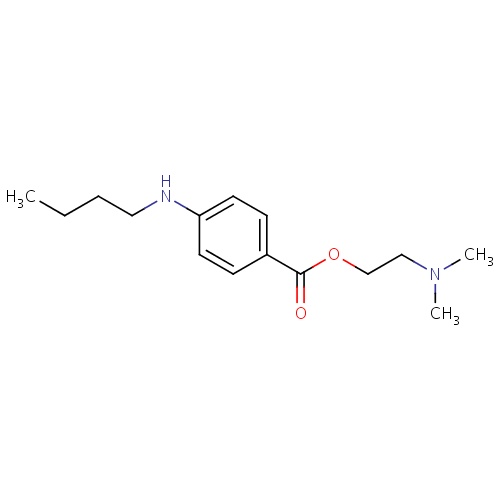
| FDBD01668 | Tetracaine |
|
Anesthetics, Local; Anesthetics; Ophthalmologicals; Sensory Organs; Dermatologicals; Nervous System; Cardiovascular System; Local Anesthetics; Agents for Treatment of Hemorrhoids and Anal Fissures for Topical Use; Vasoprotectives; Anesthetics for Topical Use; Antipruritics, Incl. Antihistamines, Anesthetics, Etc.; Esters of Aminobenzoic Acid; | |
| FDBD01715 | Florbetaben (18F) |
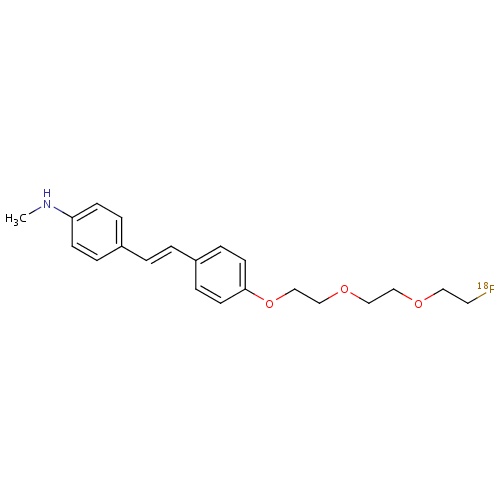
|
Diagnostic Radiopharmaceuticals; Central Nervous System; | |
| FDBD01716 | Florbetapir (18F) |
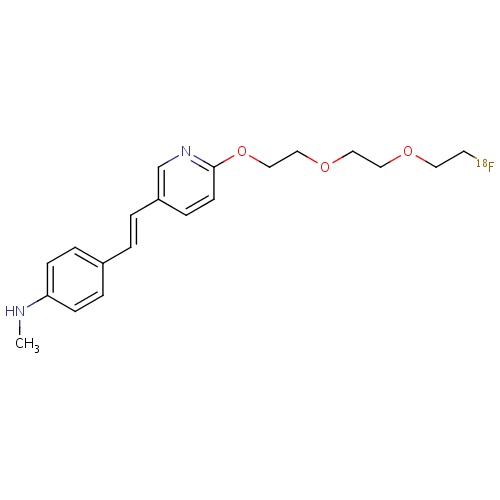
|
Diagnostic Radiopharmaceuticals; Central Nervous System; | |
| FDBD01759 | Efonidipine |
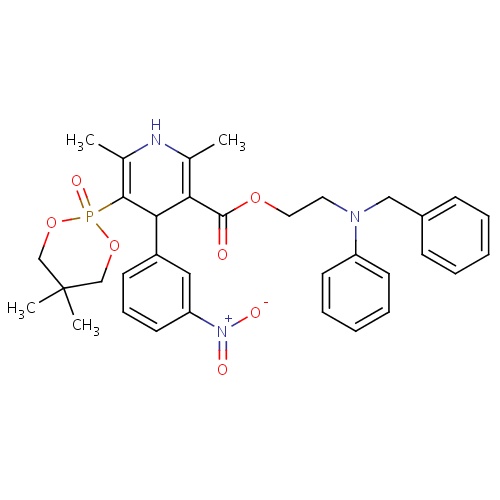
|
; | For the treatment of hypertension. |
| FDBD01810 | Osimertinib |
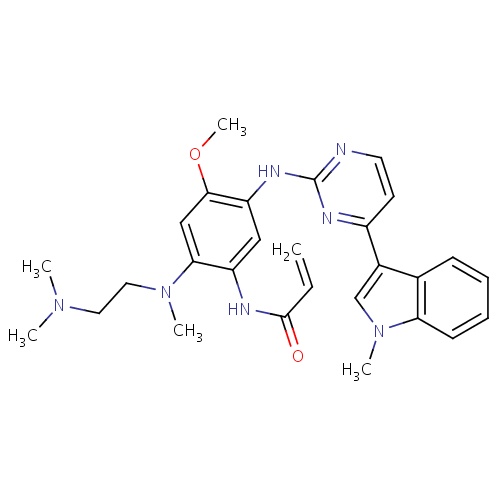
|
Antineoplastic Agents; Protein Kinase Inhibitors; Cytochrome P-450 CYP1A2 Inhibitors; Cytochrome P-450 CYP1A2 Inducers; CYP3A4 Inhibitors; | Osimertinib is indicated for the treatment of patients with metastatic epidermal growth factor receptor (EGFR) T790M mutation-positive non-small cell lung cancer (NSCLC), as detected by an FDA- approved test, who have progressed on or after EGFR-TKI therapy. |
| FDBD02441 | cambendichlor |
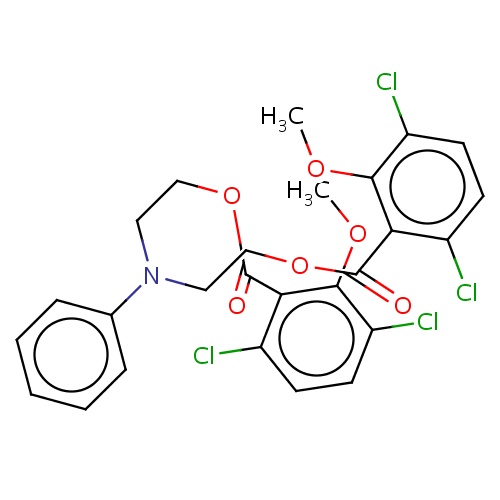
|
Herbicide | Herbicide |
| FDBD02690 | pyribambenz-isopropyl |
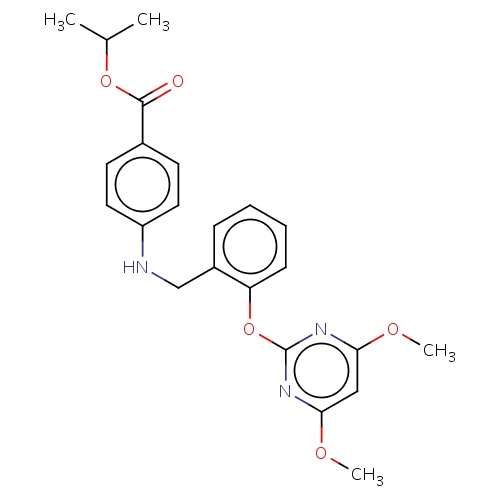
|
Herbicide | Herbicide |
| FDBD02691 | pyribambenz-propyl |
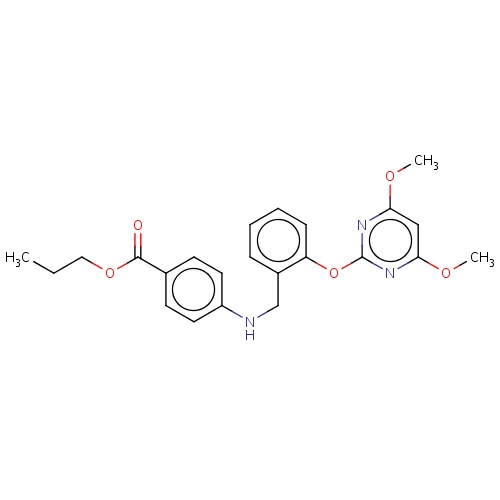
|
Herbicide | Herbicide |
22 ,
3
| FRAGNAME | PDBID | SIMILIRITY | XSCORE | SMILE | HAC |
|---|---|---|---|---|---|
| 1ci7_ligand_3_28.mol2 | 1ci7 | 1 | -6.42 | CN(C)c1ccccc1 | 9 |
| 3btc_ligand_1_4.mol2 | 3btc | 1 | -6.42 | N(C)(C)c1ccccc1 | 9 |
| 2g24_ligand_2_7.mol2 | 2g24 | 1 | -6.41 | N(c1ccccc1)C | 8 |
| 2iko_ligand_2_7.mol2 | 2iko | 1 | -6.41 | CNc1ccccc1 | 8 |
| 3btl_ligand_1_4.mol2 | 3btl | 1 | -6.40 | N(C)(C)c1ccccc1 | 9 |
| 4ocx_ligand_2_6.mol2 | 4ocx | 1 | -6.40 | N(C)(c1ccccc1)C | 9 |
| 4oyt_ligand_2_6.mol2 | 4oyt | 1 | -6.39 | CNc1ccccc1 | 8 |
| 2g2r_ligand_1_4.mol2 | 2g2r | 1 | -6.38 | c1(ccccc1)NC | 8 |
| 3eig_ligand_1_2.mol2 | 3eig | 1 | -6.37 | N(C)c1ccccc1 | 8 |
| 4hj2_ligand_2_116.mol2 | 4hj2 | 1 | -6.37 | CNc1ccccc1 | 8 |
120 ,
13

