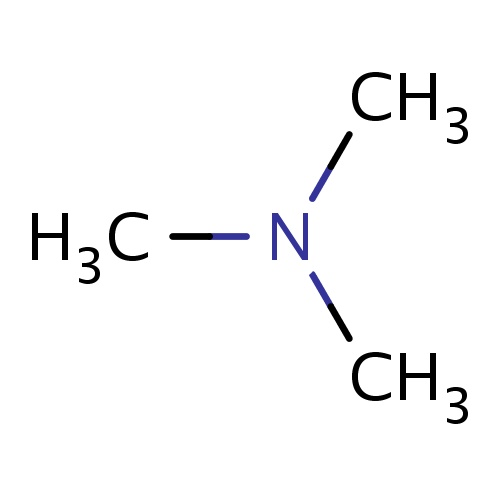
Common name
N,N-dimethylmethanamine
IUPAC name
N,N-dimethylmethanamine
SMILES
N(C)(C)C
Common name
N,N-dimethylmethanamine
IUPAC name
N,N-dimethylmethanamine
SMILES
N(C)(C)C
INCHI
InChI=1S/C3H9N/c1-4(2)3/h1-3H3
FORMULA
C3H9N

Common name
N,N-dimethylmethanamine
IUPAC name
N,N-dimethylmethanamine
Molecular weight
59.110
clogP
-0.577
clogS
-0.200
Frequency
0.0371
HBond Acceptor
1
HBond Donor
0
Total PolarSurface Area
3.24
Number of Rings
0
Rotatable Bond
0
| Drug ID | Common name | Structure CAS | Compound class | Therapeutic area |
|---|---|---|---|---|
| FDBD00406 | Toremifene |
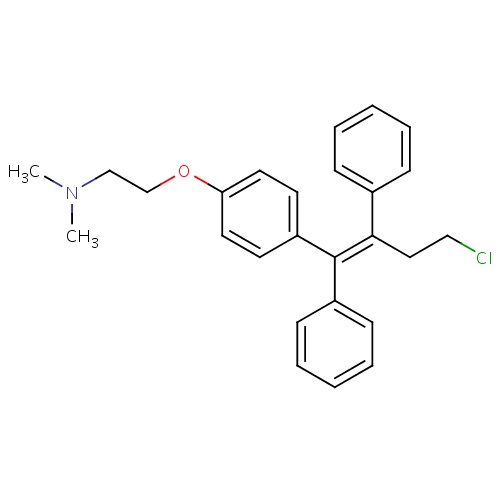
|
Antineoplastic Agents, Hormonal; Bone Density Conservation Agents; Selective Estrogen Receptor Modulators; Antineoplastic and Immunomodulating Agents; Endocrine Therapy; Hormone Antagonists and Related Agents; Anti-Estrogens; Cytochrome P-450 CYP1A2 Inhibitors; Cytochrome P-450 CYP1A2 Inducers; CYP3A4 Inhibitors; | For the treatment of metastatic breast cancer in postmenopausal women with estrogen receptor-positive or receptor-unknown tumors. Toremifene is currently under investigation as a preventative agent for prostate cancer in men with high-grade prostatic intraepithelial neoplasia and no evidence of prostate cancer. |
| FDBD00413 | Adinazolam |
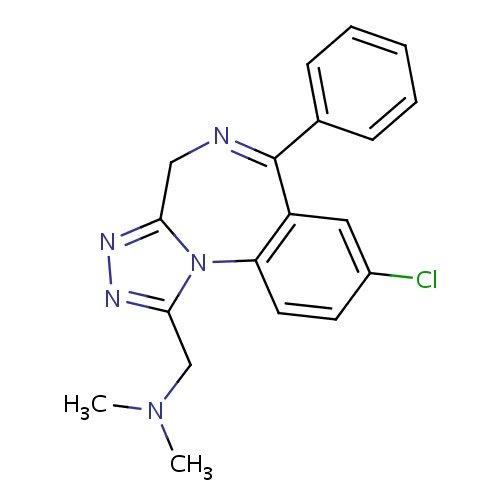
|
Antidepressive Agents; Nervous System; Benzodiazepine Derivatives; Anxiolytics; Psycholeptics; Cytochrome P-450 CYP2C19 Inducers; CYP3A4 Inhibitors; | For the treatment of anxiety and status epilepticus. |
| FDBD00448 | L-Carnitine |
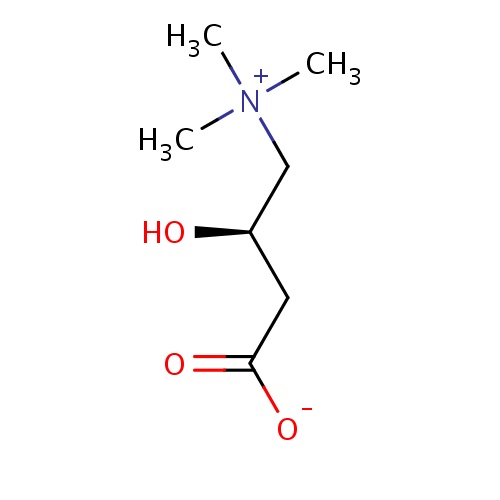
|
Vitamin B Complex; Dietary Supplements; Alimentary Tract and Metabolism; Amino Acids and Derivatives; | For treatment of primary systemic carnitine deficiency, a genetic impairment of normal biosynthesis or utilization of levocarnitine from dietary sources, or for the treatment of secondary carnitine deficiency resulting from an inborn error of metabolism such as glutaric aciduria II, methyl malonic aciduria, propionic acidemia, and medium chain fatty acylCoA dehydrogenase deficiency. Used therapeutically to stimulate gastric and pancreatic secretions and in the treatment of hyperlipoproteinemias. Parenteral levocarnitine is indicated for the prevention and treatment of carnitine deficiency in patients with end-stage renal disease. |
| FDBD00450 | Nizatidine |
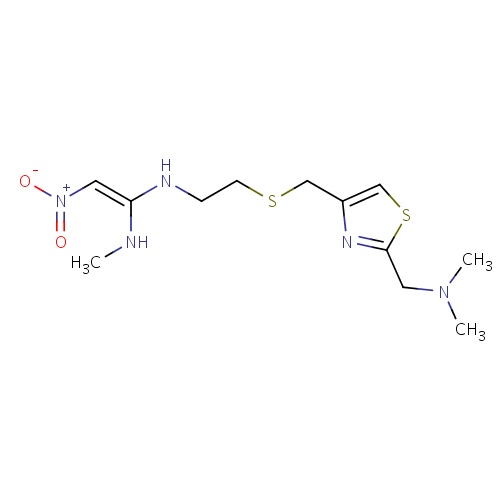
|
Anti-Ulcer Agents; Alimentary Tract and Metabolism; Drugs for Peptic Ulcer and Gastro-Oesophageal Reflux Disease (Gord); Drugs for Acid Related Disorders; H2 Antagonists; | For the treatment of acid-reflux disorders (GERD), peptic ulcer disease, active benign gastric ulcer, and active duodenal ulcer. |
| FDBD00487 | Nicardipine |
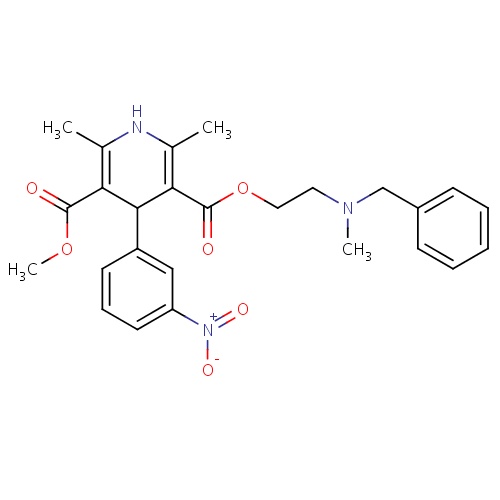
|
Antihypertensive Agents; Anti-Arrhythmia Agents; Vasodilator Agents; Calcium Channel Blockers; Adrenergic alpha-1 Receptor Antagonists; Dihydropyridines; Cardiovascular System; Dihydropyridine Derivatives; Selective Calcium Channel Blockers With Mainly Vascular Effects; Cytochrome P-450 CYP2C9 Inhibitors; Cytochrome P-450 CYP2C8 Inhibitors; Cytochrome P-450 CYP2C9 Inducers; Cytochrome P-450 CYP2C19 Inducers; Cytochrome P-450 CYP2C8 Inducers; Cytochrome P-450 CYP2B6 Inducers; Cytochrome P-450 CYP2B6 Inhibitors; CYP2B6 Inhibitors (strong); CYP2D6 Inducers; CYP2D6 Inducers (strong); CYP2E1 Inhibitors; CYP2E1 Inducers; CYP2E1 Inducers (strong); CYP3A4 Inhibitors; Combined Inhibitors of CYP3A4 and P-glycoprotein; | Used for the management of patients with chronic stable angina and for the treatment of hypertension. |
| FDBD00512 | Dextropropoxyphene |
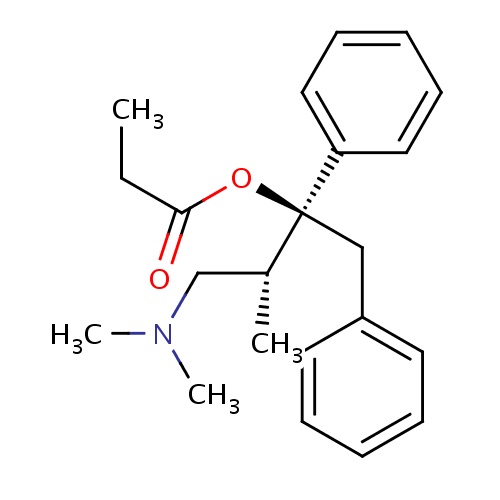
|
Analgesics; Analgesics, Opioid; Narcotics; Antitussive Agents; Nervous System; Opioids; Diphenylpropylamine Derivatives; Cytochrome P-450 CYP2C9 Inhibitors; Cytochrome P-450 CYP2C8 Inhibitors; Cytochrome P-450 CYP2C9 Inducers; Cytochrome P-450 CYP2C8 Inducers; CYP2D6 Inducers; CYP2D6 Inducers (strong); CYP3A4 Inhibitors; | For the relief of mild to moderate pain. |
| FDBD00526 | Verapamil |
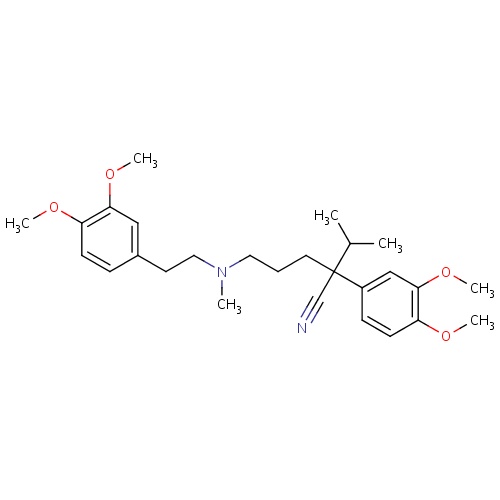
|
Anti-Arrhythmia Agents; Vasodilator Agents; Calcium Channel Blockers; Cardiovascular System; Agents Acting on the Renin-Angiotensin System; ACE Inhibitors and Calcium Channel Blockers; Selective Calcium Channel Blockers With Direct Cardiac Effects; Phenylalkylamine Derivatives; Cytochrome P-450 CYP1A2 Inhibitors; Cytochrome P-450 CYP2C9 Inhibitors; Cytochrome P-450 CYP1A2 Inducers; Cytochrome P-450 CYP2C8 Inhibitors; Cytochrome P-450 CYP2C9 Inducers; Cytochrome P-450 CYP2C19 Inducers; Cytochrome P-450 CYP2C8 Inducers; Cytochrome P-450 CYP2B6 Inducers; Cytochrome P-450 CYP2B6 Inhibitors; CYP2B6 Inhibitors (strong); CYP2D6 Inducers; CYP2D6 Inducers (strong); CYP3A4 Inhibitors; BSEP/ABCB11 Inhibitors; Combined Inhibitors of CYP3A4 and P-glycoprotein; | For the treatment of hypertension, angina, and cluster headache prophylaxis. |
| FDBD00527 | Trimethobenzamide |
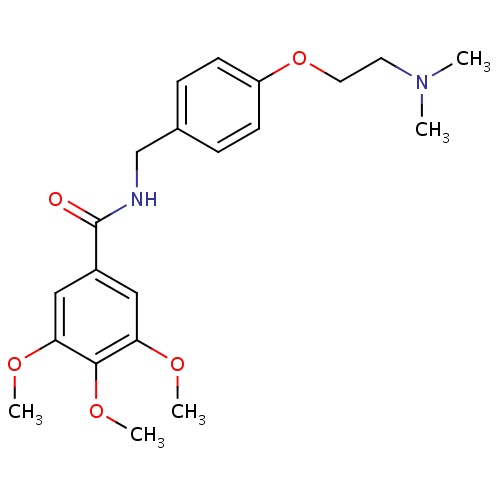
|
Antiemetics; Respiratory System; Aminoalkyl Ethers; Antihistamines for Systemic Use; | For the treatment of postoperative nausea and vomiting and for nausea associated with gastroenteritis. |
| FDBD00534 | Sumatriptan |

|
Vasoconstrictor Agents; Analgesics; Serotonin Antagonists; Serotonin 5-HT1 Receptor Agonists; Serotonin Receptor Agonists; Nervous System; Selective Serotonin (5Ht1) Agonists; Antimigraine Preparations; | For the treatment of migraine attacks with or without aura. |
| FDBD00540 | Tamoxifen |
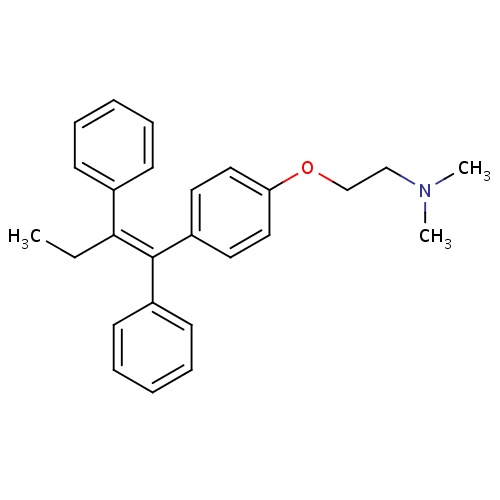
|
Antineoplastic Agents, Hormonal; Bone Density Conservation Agents; Estrogen Antagonists; Selective Estrogen Receptor Modulators; Antineoplastic and Immunomodulating Agents; Endocrine Therapy; Hormone Antagonists and Related Agents; Anti-Estrogens; Cytochrome P-450 CYP1A2 Inhibitors; Cytochrome P-450 CYP2C9 Inhibitors; Cytochrome P-450 CYP1A2 Inducers; Cytochrome P-450 CYP2C8 Inhibitors; Cytochrome P-450 CYP2C9 Inducers; Cytochrome P-450 CYP2C19 Inducers; Cytochrome P-450 CYP2C8 Inducers; Cytochrome P-450 CYP2B6 Inducers; Cytochrome P-450 CYP2B6 Inhibitors; CYP2A6 Inhibitors; CYP2A6 Inhibitors (strong); CYP2A6 Inhibitors (moderate); CYP2A6 Inducers; CYP2A6 Inducers (strong); CYP2B6 Inhibitors (strong); CYP2D6 Inducers; CYP2D6 Inducers (strong); CYP2E1 Inhibitors; CYP2E1 Inducers; CYP2E1 Inducers (strong); CYP3A4 Inhibitors; BSEP/ABCB11 Inhibitors; Combined Inducers of CYP3A4 and P-glycoprotein; Combined Inhibitors of CYP3A4 and P-glycoprotein; | Tamoxifen is indicated for the treatment of metastatic breast cancer in women and men and ductal carcinoma in Situ. |
108 ,
11
| FRAGNAME | PDBID | SIMILIRITY | XSCORE | SMILE | HAC |
|---|---|---|---|---|---|
| 2reg_ligand_1_1.mol2 | 2reg | 1 | -5.84 | C[N+](C)(C)C | 5 |
| 2rin_ligand_1_0.mol2 | 2rin | 1 | -5.74 | C[N+](C)(C)C | 5 |
| 4bgk_ligand_1_3.mol2 | 4bgk | 1 | -5.64 | [N+](C)(C)(C)C | 5 |
| 4c5w_ligand_1_0.mol2 | 4c5w | 1 | -5.64 | C[N+](C)(C)C | 5 |
| 1sw2_ligand_frag_0.mol2 | 1sw2 | 1 | -5.63 | [NH+](C)(C)C | 4 |
892 ,
90

