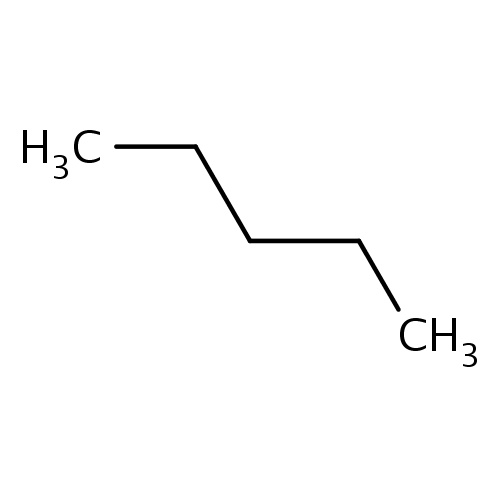
Common name
pentane
IUPAC name
pentane
SMILES
CCCCC
Common name
pentane
IUPAC name
pentane
SMILES
CCCCC
INCHI
InChI=1S/C5H12/c1-3-5-4-2/h3-5H2,1-2H3
FORMULA
C5H12

Common name
pentane
IUPAC name
pentane
Molecular weight
72.149
clogP
1.336
clogS
-1.729
Frequency
0.0316
HBond Acceptor
0
HBond Donor
0
Total PolarSurface Area
0
Number of Rings
0
Rotatable Bond
2
| Drug ID | Common name | Structure CAS | Compound class | Therapeutic area |
|---|---|---|---|---|
| FDBD00275 | Carisoprodol |
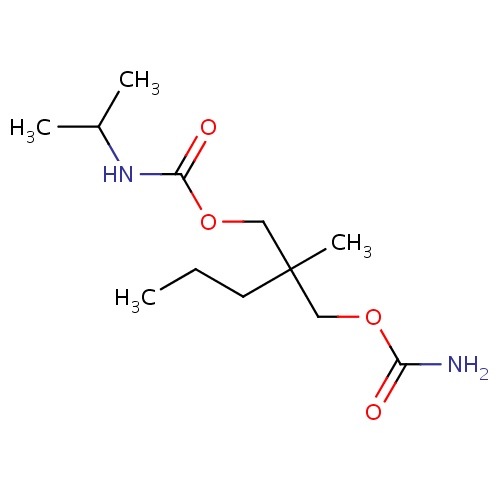
|
Muscle Relaxants, Central; Neuromuscular Agents; Muscle Relaxants, Skeletal; Musculo-Skeletal System; Muscle Relaxants, Centrally Acting Agents; Muscle Relaxants; Carbamic Acid Esters; Cytochrome P-450 CYP2C19 Inducers; | For the relief of discomfort associated with acute, painful, musculoskeletal conditions. |
| FDBD00289 | Mupirocin |
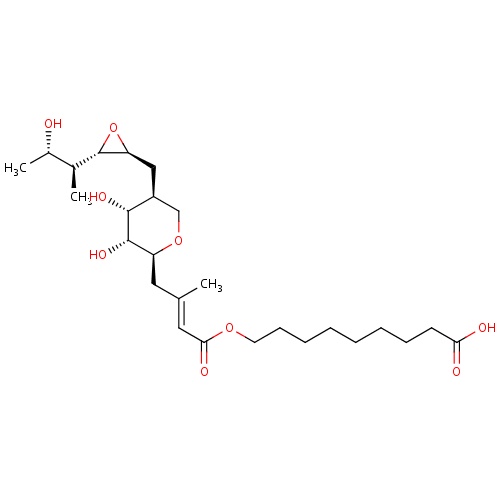
|
Anti-Bacterial Agents; Protein Synthesis Inhibitors; Antibiotics; Respiratory System; Dermatologicals; Nasal Preparations; Antibiotics for Topical Use; | For the treatment of . |
| FDBD00295 | Secobarbital |
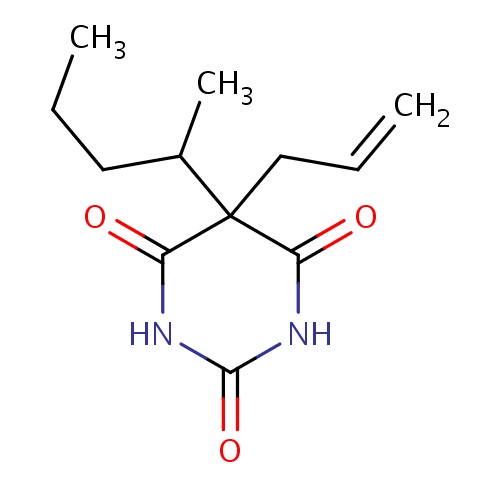
|
Hypnotics and Sedatives; Adjuvants, Anesthesia; GABA Modulators; Barbiturates; Adjuvants; Nervous System; Psycholeptics; Barbiturates, Plain; Products for Animal Euthanasia; Cytochrome P-450 CYP1A2 Inhibitors; Cytochrome P-450 CYP2C9 Inhibitors; Cytochrome P-450 CYP1A2 Inducers; Cytochrome P-450 CYP2C8 Inhibitors; Cytochrome P-450 CYP2C9 Inducers; Cytochrome P-450 CYP2C8 Inducers; | For the Short-term treatment of intractable insomnia for patients habituated to barbiturates. |
| FDBD00306 | Carboprost Tromethamine |
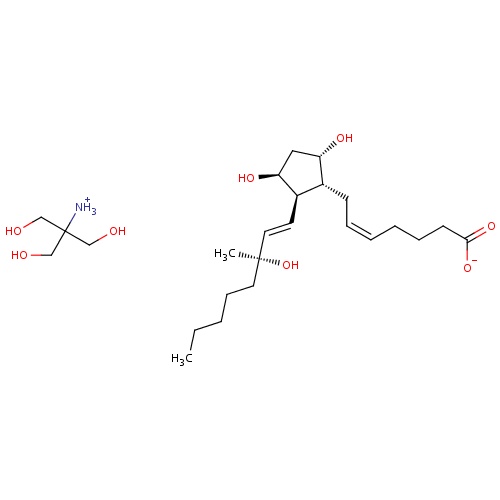
|
Oxytocics; Abortifacient Agents, Nonsteroidal; Prostaglandins; Genito Urinary System and Sex Hormones; Uterotonics; Tromethamine; | For aborting pregnancy between the 13th and 20th weeks of gestation as calculated from the first day of the last normal menstrual period and in the following conditions related to second trimester abortion: 1. Failure of expulsion of the fetus during the course of treatment by another method; 2. Premature rupture of membranes in intrauterine methods with loss of drug and insufficient or absent uterine activity; 3. Requirement of a repeat intrauterine instillation of drug for expulsion of the fetus; 4. Inadvertent or spontaneous rupture of membranes in the presence of a previable fetus and absence of adequate activity for expulsion. Also for the treatment of postpartum hemorrhage due to uterine atony which has not responded to conventional methods of management. |
| FDBD00338 | Sodium Tetradecyl Sulfate |
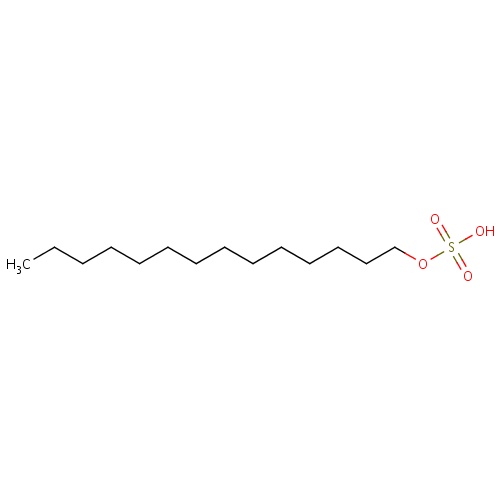
|
Sclerosing Solutions; Surface-Active Agents; Cardiovascular System; Vasoprotectives; Sclerosing Agents for Local Injection; Antivaricose Therapy; | For the treatment of small uncomplicated varicose veins of the lower extremities that show simple dilation with competent valves. |
| FDBD00343 | Dronabinol |
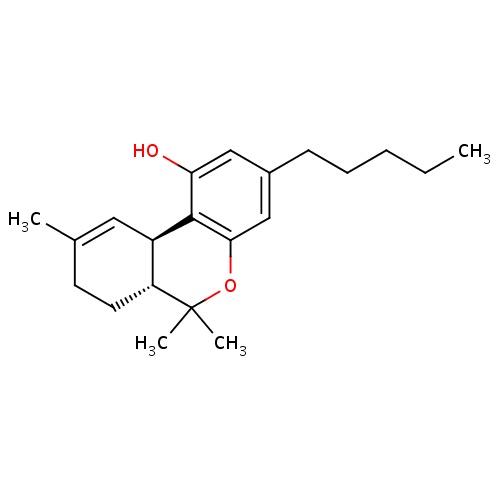
|
Analgesics, Non-Narcotic; Cannabinoid Receptor Agonists; Hallucinogens; Psychotropic Drugs; Alimentary Tract and Metabolism; Antiemetics and Antinauseants; Cytochrome P-450 CYP2C9 Inhibitors; Cytochrome P-450 CYP2C9 Inducers; Cytochrome P-450 CYP2C19 Inducers; CYP2A6 Inhibitors; CYP2A6 Inhibitors (strong); CYP2A6 Inhibitors (moderate); CYP2A6 Inducers; CYP2A6 Inducers (strong); CYP3A4 Inhibitors; | For the treatment of anorexia associated with weight loss in patients with AIDS, and nausea and vomiting associated with cancer chemotherapy in patients who have failed to respond adequately to conventional antiemetic treatments. |
| FDBD00359 | Nabilone |
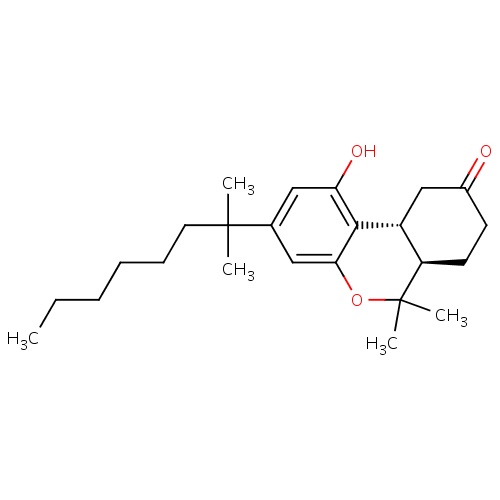
|
Alimentary Tract and Metabolism; Antiemetics and Antinauseants; | Used for the control of nausea and vomiting, caused by chemotherapeutic agents used in the treatment of cancer, in patients who have failed to respond adequately to conventional antiemetic treatments. |
| FDBD00415 | Azelaic Acid |
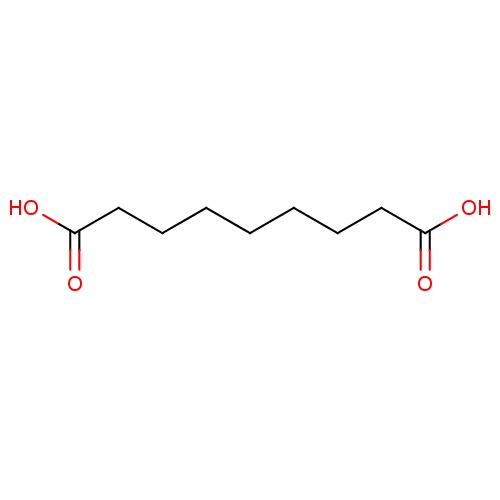
|
Antineoplastic Agents; Dermatologic Agents; Anti-Acne Preparations; Dermatologicals; Anti-Acne Preparations for Topical Use; | For the topical treatment of mild-to-moderate inflammatory acne vulgaris. |
| FDBD00464 | Thiopental |
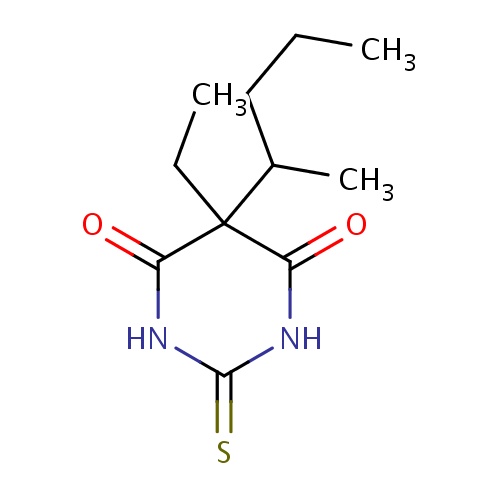
|
Hypnotics and Sedatives; Anticonvulsants; Anesthetics, Intravenous; GABA Modulators; Anesthetics; Nervous System; Psycholeptics; Anesthetics, General; Barbiturates, Plain; CYP2E1 Inhibitors; CYP2E1 Inducers; CYP2E1 Inducers (strong); CYP3A4 Inhibitors; | For use as the sole anesthetic agent for brief (15 minute) procedures, for induction of anesthesia prior to administration of other anesthetic agents, to supplement regional anesthesia, to provide hypnosis during balanced anesthesia with other agents for analgesia or muscle relaxation, for the control of convulsive states during or following inhalation anesthesia or local anesthesia, in neurosurgical patients with increased intracranial pressure, and for narcoanalysis and narcosynthesis in psychiatric disorders. |
| FDBD00473 | Chloroquine |
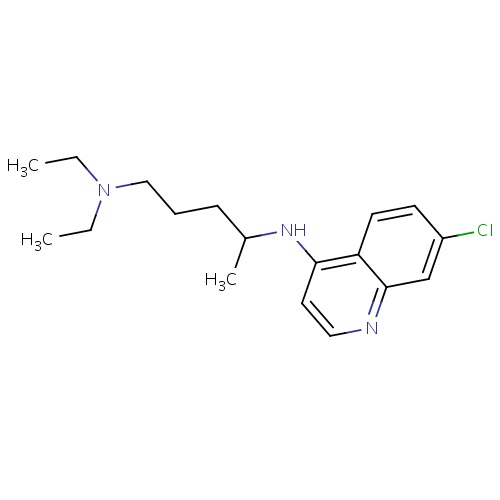
|
Antirheumatic Agents; Antimalarials; Antiprotozoal Agents; Amebicides; Antiparasitic Products, Insecticides and Repellents; Aminoquinolines; Cytochrome P-450 CYP2C8 Inhibitors; Cytochrome P-450 CYP2C8 Inducers; CYP2D6 Inducers; CYP2D6 Inducers (strong); CYP3A4 Inhibitors; | For the suppressive treatment and for acute attacks of malaria due to P. vivax, P.malariae, P. ovale, and susceptible strains of P. falciparum, Second-line agent in treatment of Rheumatoid Arthritis. |
92 ,
10
| FRAGNAME | PDBID | SIMILIRITY | XSCORE | SMILE | HAC |
|---|---|---|---|---|---|
| 4mga_ligand_2_0.mol2 | 4mga | 1 | -6.98 | C(C(C)(C)C)C(C)C | 8 |
| 1ikt_ligand_2_0.mol2 | 1ikt | 1 | -6.97 | C(C)(C)CC(C)(C)C | 8 |
| 2yiu_ligand_4_296.mol2 | 2yiu | 1 | -6.87 | C(C)(C)CC(C)C | 7 |
| 3hy5_ligand_3_31.mol2 | 3hy5 | 1 | -6.74 | CC(C)CCC | 6 |
| 1gt3_ligand_3_0.mol2 | 1gt3 | 1 | -6.73 | CCC(C)CC | 6 |
| 4u82_ligand_4_680.mol2 | 4u82 | 1 | -6.69 | CCCC(C)C | 6 |
| 5d1r_ligand_3_64.mol2 | 5d1r | 1 | -6.67 | CCCCC | 5 |
| 2igx_ligand_3_100.mol2 | 2igx | 1 | -6.63 | CCCCC | 5 |
| 2yiu_ligand_3_130.mol2 | 2yiu | 1 | -6.61 | C(C)(C)CCC | 6 |
| 2vcg_ligand_4_205.mol2 | 2vcg | 1 | -6.59 | C(C)CCC | 5 |
1224 ,
123

