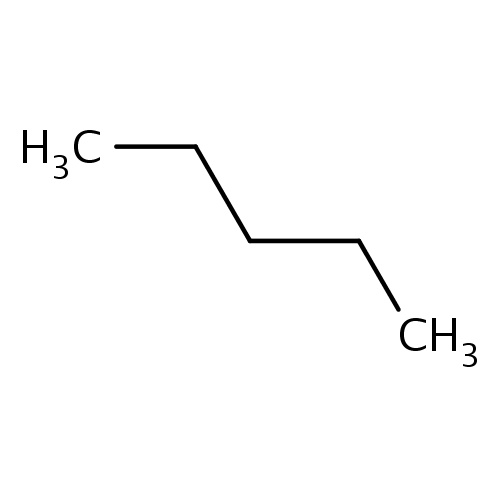
Common name
pentane
IUPAC name
pentane
SMILES
CCCCC
Common name
pentane
IUPAC name
pentane
SMILES
CCCCC
INCHI
InChI=1S/C5H12/c1-3-5-4-2/h3-5H2,1-2H3
FORMULA
C5H12

Common name
pentane
IUPAC name
pentane
Molecular weight
72.149
clogP
1.336
clogS
-1.729
Frequency
0.0316
HBond Acceptor
0
HBond Donor
0
Total PolarSurface Area
0
Number of Rings
0
Rotatable Bond
2
| Drug ID | Common name | Structure CAS | Compound class | Therapeutic area |
|---|---|---|---|---|
| FDBD01435 | Dabigatran etexilate |
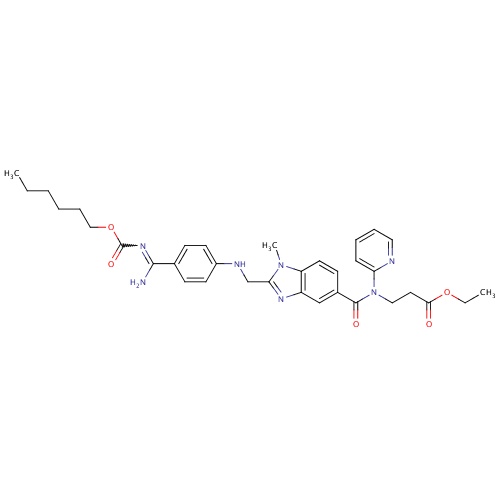
|
Antithrombins; Direct Thrombin Inhibitors; Antithrombotic Agents; Blood and Blood Forming Organs; | Dabigatran is indicated for the prevention of venous thromboembolic events in patients who have undergone elective hip or knee replacement surgery (based on RE-NOVATE, RE-MODEL, and RE-MOBILIZE trials). In 2010, it was approved in the US and Canada for prevention of stroke and systemic embolism in patients with atrial fibrillation (approval based on the RE-LY trial). Contraindications: severe renal impairment (CrCL . |
| FDBD01485 | Hydroxyprogesterone caproate |
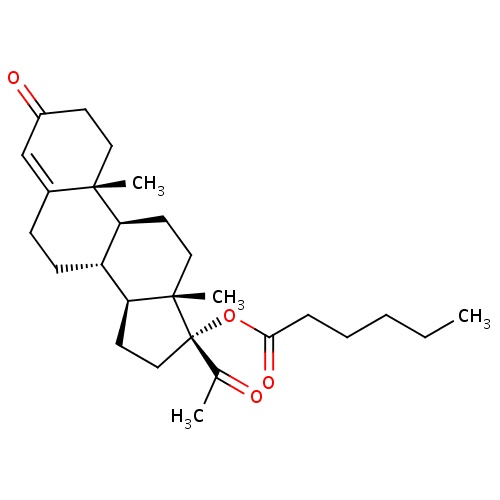
|
Sex Hormones and Modulators of the Genital System; Genito Urinary System and Sex Hormones; Progestogens; Pregnen (4) Derivatives; Cytochrome P-450 CYP2C19 Inducers; CYP3A4 Inhibitors; | Hydroxyprogesterone caproate is indicated for the prevention of spontaneous preterm births in singleton pregnancies in women who have previously had a spontaneous preterm birth. (1) . |
| FDBD01522 | Nandrolone decanoate |
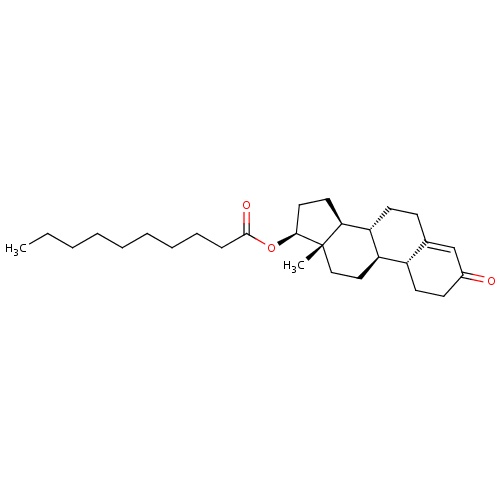
|
Anabolic Agents; | For the treatment of refractory deficient red cell production anemias, breast carcinoma, hereditary angioedema, antithrombin III deficiency, fibrinogen excess, growth failure and Turner's syndrome. It is also indicated in the prophylaxis of hereditary angioedema. |
| FDBD01548 | Fingolimod |

|
Immunosuppressive Agents; Antineoplastic and Immunomodulating Agents; Selective Immunosuppressants; CYP2D6 Inducers; CYP2D6 Inducers (strong); CYP2E1 Inhibitors; CYP2E1 Inducers; CYP2E1 Inducers (strong); CYP3A4 Inhibitors; | Fingolimod is a sphingosine 1-phosphate receptor modulator indicated for the treatment of patients with relapsing forms of multiple sclerosis to reduce the frequency of clinical exacerbations and to delay the accumulation of physical disability. |
| FDBD01592 | Hexoprenaline |
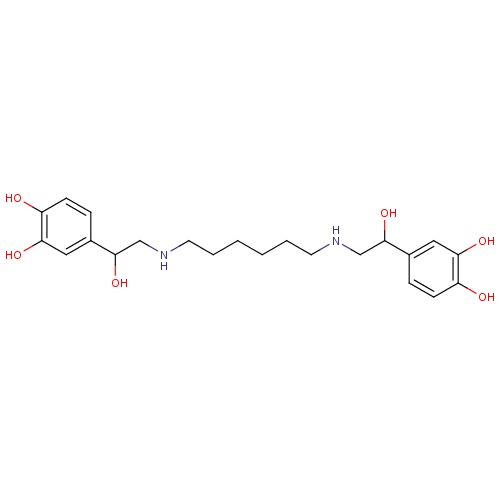
|
Adrenergic beta-2 Receptor Agonists; Bronchodilator Agents; Tocolytic Agents; Respiratory System; Drugs for Obstructive Airway Diseases; Selective Beta-2-Adrenoreceptor Agonists; Adrenergics, Inhalants; Adrenergics for Systemic Use; | |
| FDBD01593 | Hexetidine |
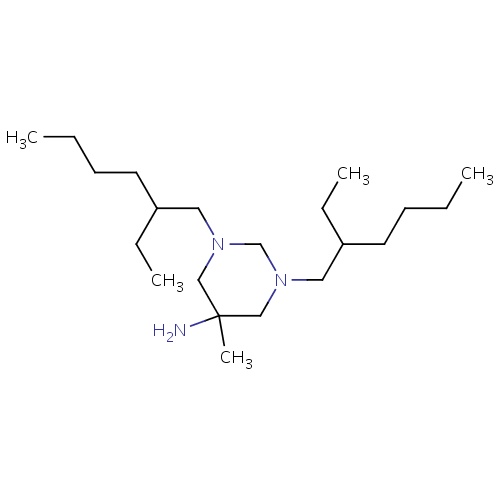
|
Antifungal Agents; Anti-Infective Agents, Local; Genito Urinary System and Sex Hormones; Alimentary Tract and Metabolism; Stomatological Preparations; Gynecological Antiinfectives and Antiseptics; Antiinfectives and Antiseptics for Local Oral Treatment; | |
| FDBD01637 | Miltefosine |

|
Antineoplastic Agents; Antineoplastic and Immunomodulating Agents; | For the treatment of mucosal (caused by Leishmania braziliensis), cutaneous (caused by L. braziliensis, L. guyanensis, and L. panamensis), and visceral leishmaniasis (caused by L. donovani). In comparing Leishmania drug susceptibility, it has been found that L. donovani is the most susceptible to miltefosine while L. major is the least susceptible. Off-label use includes treatment of free-living amebae (FLA) infections (unlabeled use; CDC, 2013). |
| FDBD01640 | Eliglustat |
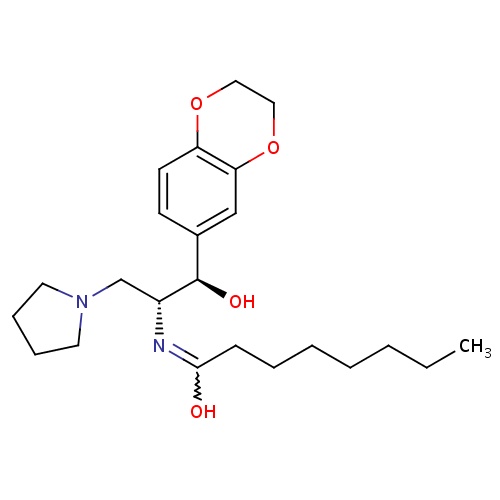
|
Enzyme Inhibitors; Alimentary Tract and Metabolism; Various Alimentary Tract and Metabolism Products; CYP2D6 Inducers; CYP2D6 Inducers (strong); CYP3A4 Inhibitors; | Eliglustat is indicated for the long-term treatment of type 1 Gaucher disease in patients who are CYP2D6 extensive metabolizers (EMs), intermediate metabolizers (IMs), or poor metabolizers (PMs) in treatment-naive and treatment-experienced adult patients. |
| FDBD01652 | Cobicistat |
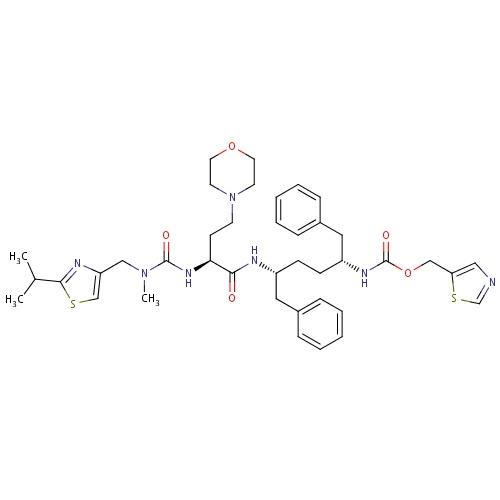
|
Anti-HIV Agents; Antiinfectives for Systemic Use; Direct Acting Antivirals; Antivirals for Systemic Use; Cytochrome P-450 CYP3A Inhibitors; CYP2D6 Inducers; CYP2D6 Inducers (strong); | Cobicistat is a CYP3A inhibitor indicated to increase systemic exposure of atazanavir or darunavir (once daily dosing regimen) in combination with other antiretroviral agents in the treatment of HIV-1 infection. It is not interchangeable with ritonavir to increase systemic exposure of darunavir 600 mg twice daily, fosamprenavir, saquinavir, or tipranavir due to lack of exposure data. The use of cobicistat is not recommended with darunavir 600 mg twice daily, fosamprenavir, saquinavir or tipranavir. Complex or unknown mechanisms of drug interactions preclude extrapolation of ritonavir drug interactions to certain cobicistat interactions. Cobicistat and ritonavir when administered with either atazanavir or darunavir may result in different drug interactions when used with concomitant medications. |
| FDBD01664 | Idebenone |

|
Nervous System; Psychoanaleptics; Psychostimulants, Agents Used for Adhd and Nootropics; |
92 ,
10
| FRAGNAME | PDBID | SIMILIRITY | XSCORE | SMILE | HAC |
|---|---|---|---|---|---|
| 4mga_ligand_2_0.mol2 | 4mga | 1 | -6.98 | C(C(C)(C)C)C(C)C | 8 |
| 1ikt_ligand_2_0.mol2 | 1ikt | 1 | -6.97 | C(C)(C)CC(C)(C)C | 8 |
| 2yiu_ligand_4_296.mol2 | 2yiu | 1 | -6.87 | C(C)(C)CC(C)C | 7 |
| 3hy5_ligand_3_31.mol2 | 3hy5 | 1 | -6.74 | CC(C)CCC | 6 |
| 1gt3_ligand_3_0.mol2 | 1gt3 | 1 | -6.73 | CCC(C)CC | 6 |
| 4u82_ligand_4_680.mol2 | 4u82 | 1 | -6.69 | CCCC(C)C | 6 |
| 5d1r_ligand_3_64.mol2 | 5d1r | 1 | -6.67 | CCCCC | 5 |
| 2igx_ligand_3_100.mol2 | 2igx | 1 | -6.63 | CCCCC | 5 |
| 2yiu_ligand_3_130.mol2 | 2yiu | 1 | -6.61 | C(C)(C)CCC | 6 |
| 2vcg_ligand_4_205.mol2 | 2vcg | 1 | -6.59 | C(C)CCC | 5 |
1224 ,
123

