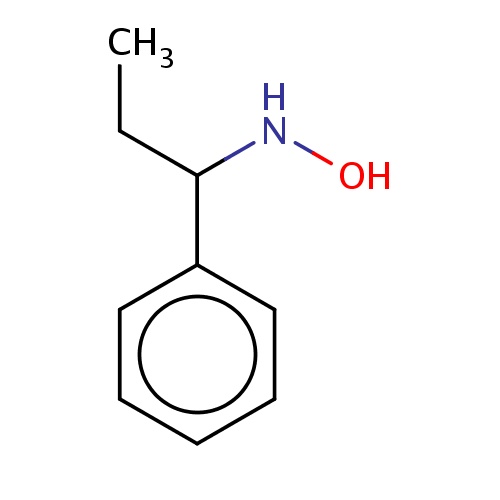
Common name
N-[(1S)-1-phenylpropyl]hydroxylamine
IUPAC name
N-[(1S)-1-phenylpropyl]hydroxylamine
SMILES
CCC(c1ccccc1)NO
Common name
N-[(1S)-1-phenylpropyl]hydroxylamine
IUPAC name
N-[(1S)-1-phenylpropyl]hydroxylamine
SMILES
CCC(c1ccccc1)NO
INCHI
InChI=1S/C9H13NO/c1-2-9(10-11)8-6-4-3-5-7-8/h3-7,9-11H,2H2,1H3
FORMULA
C9H13NO

Common name
N-[(1S)-1-phenylpropyl]hydroxylamine
IUPAC name
N-[(1S)-1-phenylpropyl]hydroxylamine
Molecular weight
151.206
clogP
0.779
clogS
-1.939
Frequency
0.0003
HBond Acceptor
1
HBond Donor
2
Total PolarSurface Area
20.23
Number of Rings
1
Rotatable Bond
3
| Drug ID | Common name | Structure CAS | Compound class | Therapeutic area |
|---|---|---|---|---|
| FDBD00065 | Fluvoxamine |
|
Antidepressive Agents, Second-Generation; Anti-Anxiety Agents; Serotonin Uptake Inhibitors; Nervous System; Antidepressants; Psychoanaleptics; Selective Serotonin Reuptake Inhibitors; Cytochrome P-450 CYP1A2 Inhibitors; Cytochrome P-450 CYP2C9 Inhibitors; Cytochrome P-450 CYP1A2 Inducers; Cytochrome P-450 CYP2C9 Inducers; Cytochrome P-450 CYP2C19 Inducers; Cytochrome P-450 CYP2B6 Inducers; Cytochrome P-450 CYP2B6 Inhibitors; CYP2B6 Inhibitors (strong); CYP2D6 Inducers; CYP2D6 Inducers (strong); CYP2E1 Inhibitors; CYP2E1 Inducers; CYP2E1 Inducers (strong); CYP3A4 Inhibitors; Combined Inhibitors of CYP3A4 and P-glycoprotein; | For management of depression and for Obsessive Compulsive Disorder (OCD). Has also been used in the management of bulimia nervosa. |
1 ,
1
| FRAGNAME | PDBID | SIMILIRITY | XSCORE | SMILE | HAC |
|---|---|---|---|---|---|
| 4mm9_ligand_4_175.mol2 | 4mm9 | 1 | -7.43 | O[NH2+][C@H](CC)c1ccccc1 | 11 |
| 4mm9_ligand_3_100.mol2 | 4mm9 | 0.828571 | -7.14 | O[NH2+][C@H](C)c1ccccc1 | 10 |
| 4mm9_ligand_3_85.mol2 | 4mm9 | 0.8 | -7.31 | [C@@H]([NH3+])(CC)c1ccccc1 | 10 |
| 4uvc_ligand_3_814.mol2 | 4uvc | 0.8 | -6.91 | CC[C@H](c1ccccc1)[NH3+] | 10 |
| 4mm9_ligand_4_195.mol2 | 4mm9 | 0.725 | -7.24 | CO[NH2+][C@H](C)c1ccccc1 | 11 |
| 4mm9_ligand_4_140.mol2 | 4mm9 | 0.717949 | -7.47 | [C@@H]([NH3+])(CCC)c1ccccc1 | 11 |
| 2yit_ligand_2_7.mol2 | 2yit | 0.717949 | -7.30 | c1ccc(cc1)[C@@H]([NH3+])CCC | 11 |
| 4mm9_ligand_2_35.mol2 | 4mm9 | 0.685714 | -6.77 | O[NH2+]Cc1ccccc1 | 9 |
| 4zh2_ligand_frag_1.mol2 | 4zh2 | 0.685714 | -6.45 | c1cc(ccc1)C[NH2+]O | 9 |
| 2nv7_ligand_1_0.mol2 | 2nv7 | 0.666667 | -7.86 | c1ccc2c(c1)c(ccc2)C[NH2+]O | 13 |
300 ,
31

