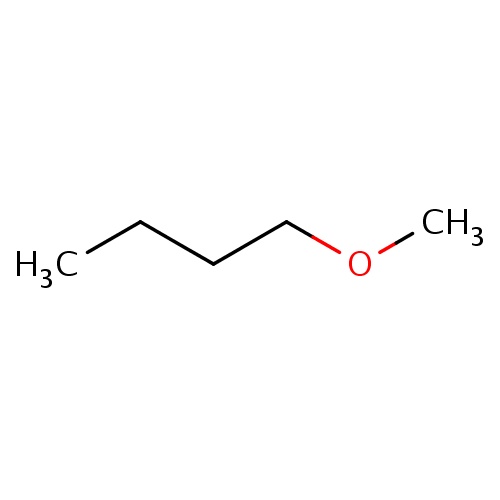
Common name
1-methoxybutane
IUPAC name
1-methoxybutane
SMILES
C(CCC)OC
Common name
1-methoxybutane
IUPAC name
1-methoxybutane
SMILES
C(CCC)OC
INCHI
InChI=1S/C5H12O/c1-3-4-5-6-2/h3-5H2,1-2H3
FORMULA
C5H12O

Common name
1-methoxybutane
IUPAC name
1-methoxybutane
Molecular weight
88.148
clogP
1.001
clogS
-1.507
Frequency
0.0027
HBond Acceptor
1
HBond Donor
0
Total PolarSurface Area
9.23
Number of Rings
0
Rotatable Bond
3
| Drug ID | Common name | Structure CAS | Compound class | Therapeutic area |
|---|---|---|---|---|
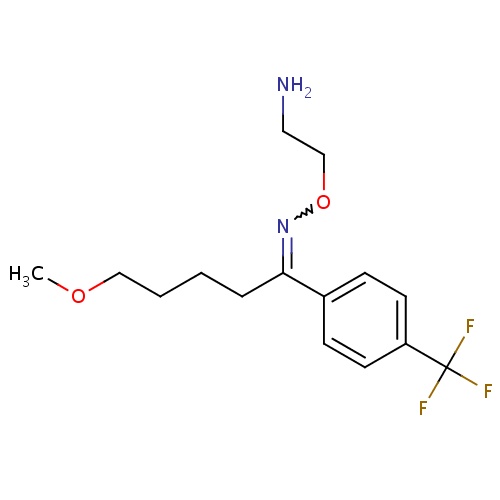
| FDBD00065 | Fluvoxamine |
|
Antidepressive Agents, Second-Generation; Anti-Anxiety Agents; Serotonin Uptake Inhibitors; Nervous System; Antidepressants; Psychoanaleptics; Selective Serotonin Reuptake Inhibitors; Cytochrome P-450 CYP1A2 Inhibitors; Cytochrome P-450 CYP2C9 Inhibitors; Cytochrome P-450 CYP1A2 Inducers; Cytochrome P-450 CYP2C9 Inducers; Cytochrome P-450 CYP2C19 Inducers; Cytochrome P-450 CYP2B6 Inducers; Cytochrome P-450 CYP2B6 Inhibitors; CYP2B6 Inhibitors (strong); CYP2D6 Inducers; CYP2D6 Inducers (strong); CYP2E1 Inhibitors; CYP2E1 Inducers; CYP2E1 Inducers (strong); CYP3A4 Inhibitors; Combined Inhibitors of CYP3A4 and P-glycoprotein; | For management of depression and for Obsessive Compulsive Disorder (OCD). Has also been used in the management of bulimia nervosa. |
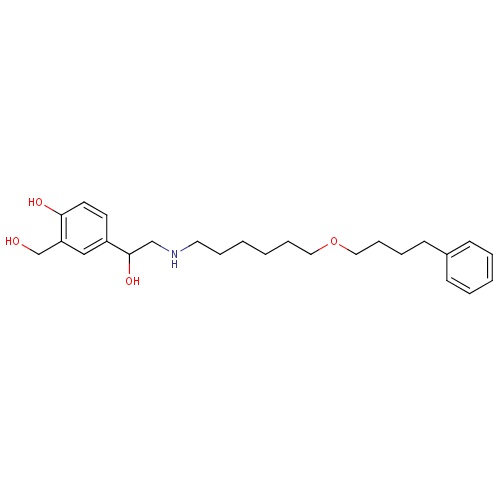
| FDBD00793 | Salmeterol |
|
Sympathomimetics; Adrenergic beta-2 Receptor Agonists; Bronchodilator Agents; Respiratory System; Drugs for Obstructive Airway Diseases; Selective Beta-2-Adrenoreceptor Agonists; Adrenergics, Inhalants; Cytochrome P-450 CYP2C8 Inhibitors; Cytochrome P-450 CYP2C8 Inducers; CYP3A4 Inhibitors; Beta2 Agonists; | For the treatment of asthma and chronic obstructive pulmonary disease (COPD). |
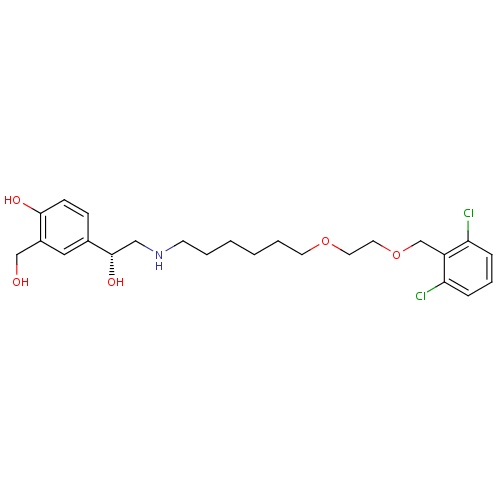
| FDBD01665 | Vilanterol |
|
Immunosuppressive Agents; Respiratory System; Drugs for Obstructive Airway Diseases; Adrenergics, Inhalants; CYP3A4 Inhibitors; Beta2 Agonists; | Vilanterol is approved for use in several combination products such as with fluticasone furoate under the tradename Breo Ellipta and in combination with umeclidinium bromide as Anoro Ellipta. Approved by the FDA in 2013, use of Breo Ellipta is indicated for the long-term, once-daily maintenance treatment of airflow obstruction in patients with COPD, including chronic bronchitis and emphysema. It is also indicated for once-daily maintenance treatment of asthma in patients aged 18 or older with reversible obstructive airways disease. |
| FDBD01822 | Piperonyl butoxide |
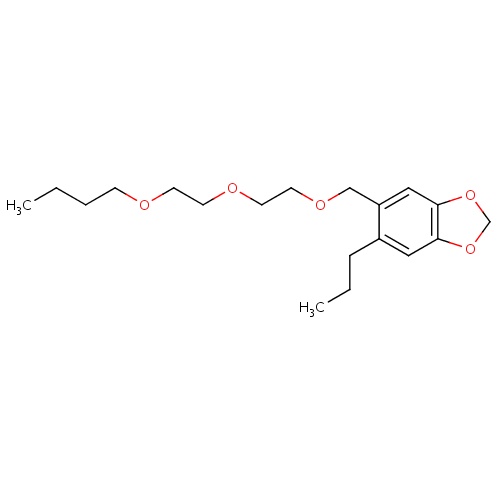
|
Pesticide Synergists; | For the treatment of head, pubic (crab), and body lice. |
| FDBD01831 | Selexipag |
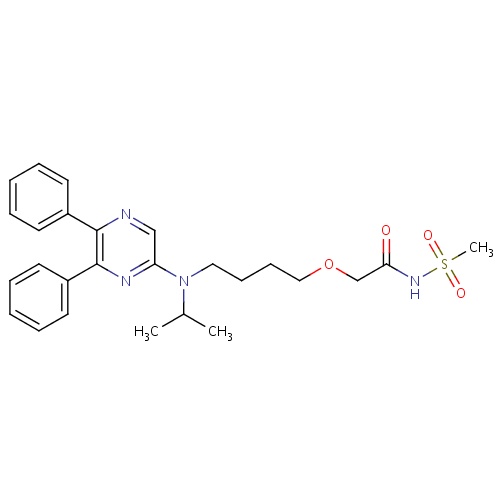
|
Antihypertensive Agents; Antithrombotic Agents; Blood and Blood Forming Organs; Platelet Aggregation Inhibitors Excl. Heparin; Cytochrome P-450 CYP2C8 Inhibitors; Cytochrome P-450 CYP2C8 Inducers; CYP3A4 Inhibitors; | Selexipag is indicated for the treatment of pulmonary arterial hypertension (PAH) to delay disease progression and reduce risk of hospitalization. |
| FDBD02409 | butachlor |
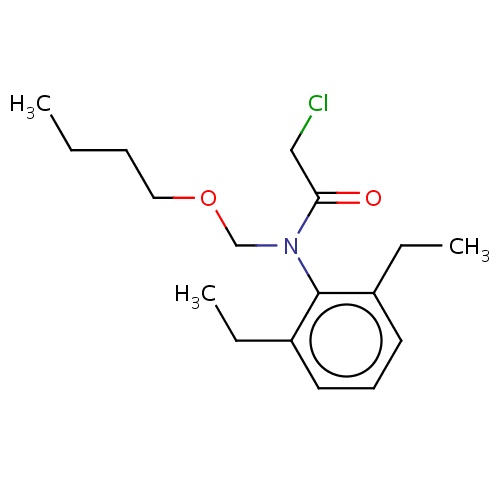
|
Herbicide | Herbicide |
| FDBD02422 | terbuchlor |
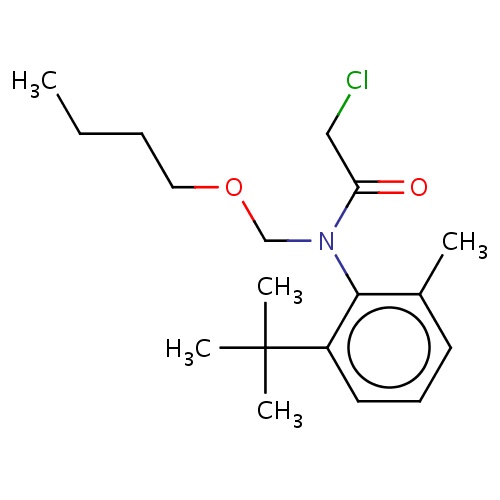
|
Herbicide | Herbicide |
| FDBD03173 | izopamfos |
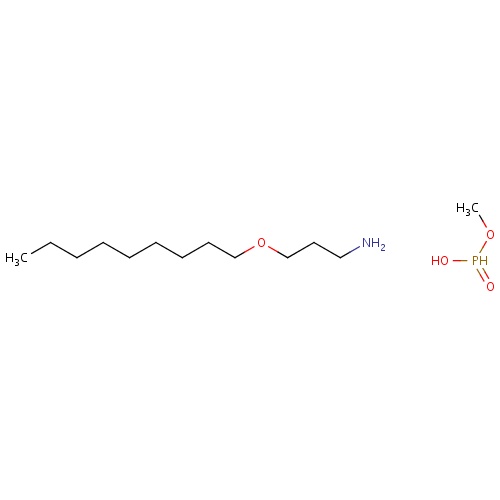
|
Fungicide | Fungicide |
8 ,
1
| FRAGNAME | PDBID | SIMILIRITY | XSCORE | SMILE | HAC |
|---|---|---|---|---|---|
| 2yiu_ligand_4_261.mol2 | 2yiu | 1 | -6.18 | CC[C@H](C)COC | 7 |
| 3gw5_ligand_4_140.mol2 | 3gw5 | 1 | -5.95 | C(OC)CCC | 6 |
| 4gjd_ligand_4_209.mol2 | 4gjd | 1 | -5.92 | O(C)CCCC | 6 |
| 4k43_ligand_4_931.mol2 | 4k43 | 1 | -5.77 | CC[C@H](C)COC | 7 |
| 4mm9_ligand_4_0.mol2 | 4mm9 | 1 | -5.68 | CCCCOC | 6 |
| 4dv8_ligand_4_145.mol2 | 4dv8 | 1 | -5.67 | CCCCOC | 6 |
| 2y8c_ligand_5_450.mol2 | 2y8c | 1 | -5.27 | C(COC)CC | 6 |
107 ,
11

