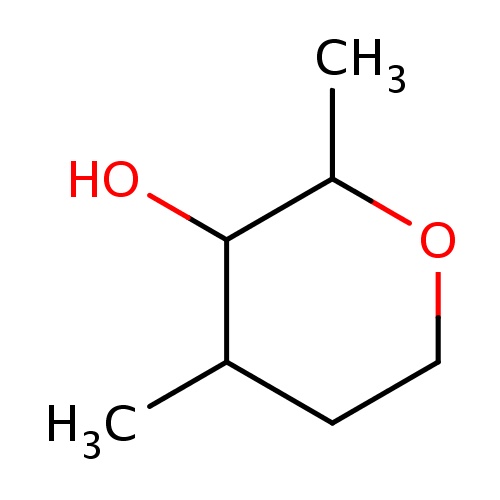
Common name
(2R,3S,4S)-2,4-dimethyltetrahydropyran-3-ol
IUPAC name
(2R,3S,4S)-2,4-dimethyltetrahydropyran-3-ol
SMILES
O1CCC(C(C1C)O)C
Common name
(2R,3S,4S)-2,4-dimethyltetrahydropyran-3-ol
IUPAC name
(2R,3S,4S)-2,4-dimethyltetrahydropyran-3-ol
SMILES
O1CCC(C(C1C)O)C
INCHI
InChI=1S/C7H14O2/c1-5-3-4-9-6(2)7(5)8/h5-8H,3-4H2,1-2H3/t5-,6+,7-/m0/s1
FORMULA
C7H14O2

Common name
(2R,3S,4S)-2,4-dimethyltetrahydropyran-3-ol
IUPAC name
(2R,3S,4S)-2,4-dimethyltetrahydropyran-3-ol
Molecular weight
130.185
clogP
0.980
clogS
-0.479
Frequency
0.0014
HBond Acceptor
2
HBond Donor
1
Total PolarSurface Area
29.46
Number of Rings
1
Rotatable Bond
0
| Drug ID | Common name | Structure CAS | Compound class | Therapeutic area |
|---|---|---|---|---|
| FDBD00087 | Erythromycin |

|
Gastrointestinal Agents; Enzyme Inhibitors; Anti-Bacterial Agents; Protein Synthesis Inhibitors; Macrolides; Anti-Acne Preparations; Antibiotics; Ophthalmologicals; Sensory Organs; Antibacterials for Systemic Use; Antiinfectives for Systemic Use; Dermatologicals; Anti-Acne Preparations for Topical Use; Antiinfectives; Antiinfectives for Treatment of Acne; Macrolides, Lincosamides and Streptogramins; Macrolides and Lincosamides for Intramammary Use; Antibacterials for Intramammary Use; Cytochrome P-450 CYP1A2 Inhibitors; Cytochrome P-450 CYP1A2 Inducers; Cytochrome P-450 CYP2B6 Inducers; Cytochrome P-450 CYP2B6 Inhibitors; CYP2B6 Inhibitors (strong); CYP3A4 Inhibitors; Combined Inhibitors of CYP3A4 and P-glycoprotein; | For use in the treatment of infections caused by susceptible strains of microorganisms in the following diseases: respiratory tract infections (upper and lower) of mild to moderate degree, pertussis (whooping cough), as adjunct to antitoxin in infections due to . |
| FDBD00095 | Azithromycin |
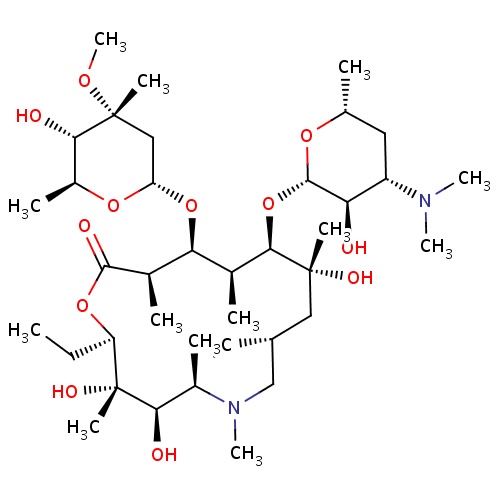
|
Anti-Bacterial Agents; Macrolides; Antibiotics; Ophthalmologicals; Sensory Organs; Antibacterials for Systemic Use; Antiinfectives for Systemic Use; Antiinfectives; Macrolides, Lincosamides and Streptogramins; Cytochrome P-450 CYP1A2 Inhibitors; Cytochrome P-450 CYP1A2 Inducers; CYP2A6 Inhibitors; CYP2A6 Inhibitors (strong); CYP2A6 Inhibitors (moderate); CYP2A6 Inducers; CYP2A6 Inducers (strong); CYP3A4 Inhibitors; Combined Inhibitors of CYP3A4 and P-glycoprotein; | For the treatment of patients with mild to moderate infections caused by susceptible strains of the designated microorganisms in the specific conditions: . |
| FDBD00641 | Roxithromycin |

|
Anti-Bacterial Agents; Macrolides; Antibacterials for Systemic Use; Antiinfectives for Systemic Use; Macrolides, Lincosamides and Streptogramins; Cytochrome P-450 CYP1A2 Inhibitors; Cytochrome P-450 CYP1A2 Inducers; Cytochrome P-450 CYP2B6 Inducers; Cytochrome P-450 CYP2B6 Inhibitors; CYP2B6 Inhibitors (strong); CYP3A4 Inhibitors; | Used to treat respiratory tract, urinary and soft tissue infections. |
| FDBD01057 | Clarithromycin |

|
Anti-Bacterial Agents; Protein Synthesis Inhibitors; Macrolides; Alimentary Tract and Metabolism; Antibacterials for Systemic Use; Antiinfectives for Systemic Use; Macrolides, Lincosamides and Streptogramins; Drugs for Peptic Ulcer and Gastro-Oesophageal Reflux Disease (Gord); Drugs for Acid Related Disorders; Cytochrome P-450 CYP1A2 Inhibitors; Cytochrome P-450 CYP1A2 Inducers; Cytochrome P-450 CYP3A Inhibitors; Cytochrome P-450 CYP2C19 Inducers; CYP3A4 Inhibitors; Combined Inhibitors of CYP3A4 and P-glycoprotein; | An alternative medication for the treatment of acute otitis media caused by . |
4 ,
1
| FRAGNAME | PDBID | SIMILIRITY | XSCORE | SMILE | HAC |
|---|---|---|---|---|---|
| 1m7d_ligand_frag_1.mol2 | 1m7d | 1 | -6.39 | C1CC[C@H]([C@@H](O1)C)O | 8 |
| 4urn_ligand_frag_2.mol2 | 4urn | 1 | -6.34 | C1C[C@H](COC1(C)C)O | 9 |
| 4uro_ligand_frag_2.mol2 | 4uro | 1 | -6.30 | C1C[C@H](COC1(C)C)O | 9 |
| 1kzn_ligand_frag_1.mol2 | 1kzn | 1 | -6.24 | C1C(OC[C@@H](C1)O)(C)C | 9 |
| 1aj6_ligand_frag_2.mol2 | 1aj6 | 1 | -6.19 | C1C(OC[C@@H](C1)O)(C)C | 9 |
| 2iyf_ligand_frag_4.mol2 | 2iyf | 1 | -5.91 | C1[C@@H](CC[C@H](O1)C)O | 8 |
| 2iyf_ligand_frag_6.mol2 | 2iyf | 1 | -5.38 | C1C[C@@H]([C@H]([C@@H](O1)C)O)C | 9 |
| 4oeg_ligand_1_3.mol2 | 4oeg | 1 | -5.12 | C[C@@H]1[C@H](CCCO1)O | 8 |
100 ,
11

