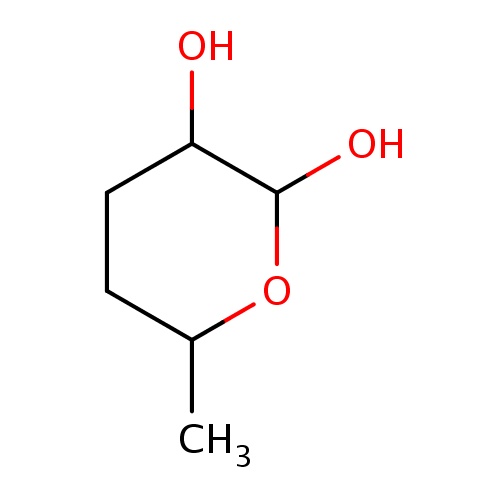
Common name
(2S,3R,6S)-6-methyltetrahydropyran-2,3-diol
IUPAC name
(2S,3R,6S)-6-methyltetrahydropyran-2,3-diol
SMILES
C1(C(OC(CC1)C)O)O
Common name
(2S,3R,6S)-6-methyltetrahydropyran-2,3-diol
IUPAC name
(2S,3R,6S)-6-methyltetrahydropyran-2,3-diol
SMILES
C1(C(OC(CC1)C)O)O
INCHI
InChI=1S/C6H12O3/c1-4-2-3-5(7)6(8)9-4/h4-8H,2-3H2,1H3/t4-,5+,6-/m0/s1
FORMULA
C6H12O3

Common name
(2S,3R,6S)-6-methyltetrahydropyran-2,3-diol
IUPAC name
(2S,3R,6S)-6-methyltetrahydropyran-2,3-diol
Molecular weight
132.158
clogP
0.061
clogS
0.481
Frequency
0.0007
HBond Acceptor
3
HBond Donor
2
Total PolarSurface Area
49.69
Number of Rings
1
Rotatable Bond
0
| Drug ID | Common name | Structure CAS | Compound class | Therapeutic area |
|---|---|---|---|---|
| FDBD00095 | Azithromycin |
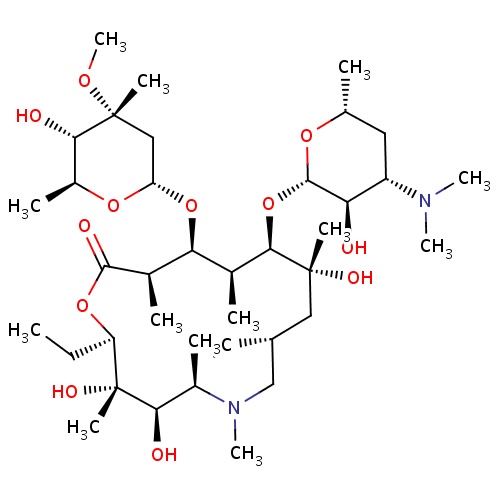
|
Anti-Bacterial Agents; Macrolides; Antibiotics; Ophthalmologicals; Sensory Organs; Antibacterials for Systemic Use; Antiinfectives for Systemic Use; Antiinfectives; Macrolides, Lincosamides and Streptogramins; Cytochrome P-450 CYP1A2 Inhibitors; Cytochrome P-450 CYP1A2 Inducers; CYP2A6 Inhibitors; CYP2A6 Inhibitors (strong); CYP2A6 Inhibitors (moderate); CYP2A6 Inducers; CYP2A6 Inducers (strong); CYP3A4 Inhibitors; Combined Inhibitors of CYP3A4 and P-glycoprotein; | For the treatment of patients with mild to moderate infections caused by susceptible strains of the designated microorganisms in the specific conditions: . |
| FDBD00809 | Dirithromycin |

|
Anti-Infective Agents; Macrolides; Antibacterials for Systemic Use; Antiinfectives for Systemic Use; Macrolides, Lincosamides and Streptogramins; | For the treatment of the following mild-to-moderate infections caused by susceptible strains of microorganisms: acute bacterial exacerbations of chronic bronchitis, secondary bacterial infection of acute bronchitis, community-acquired pneumonia, pharyngitis/tonsilitis, and uncomplicated skin and skin structure infections. |
2 ,
1
| FRAGNAME | PDBID | SIMILIRITY | XSCORE | SMILE | HAC |
|---|---|---|---|---|---|
| 4urn_ligand_1_2.mol2 | 4urn | 1 | -6.41 | O[C@H]1[C@@H](CCC(O1)(C)C)O | 10 |
| 4uro_ligand_1_2.mol2 | 4uro | 1 | -6.37 | C1C[C@H]([C@@H](OC1(C)C)O)O | 10 |
| 1kzn_ligand_1_2.mol2 | 1kzn | 1 | -6.31 | C1C(O[C@H]([C@@H](C1)O)O)(C)C | 10 |
| 1aj6_ligand_1_2.mol2 | 1aj6 | 1 | -6.25 | C1C(O[C@H]([C@@H](C1)O)O)(C)C | 10 |
| 2iyf_ligand_1_4.mol2 | 2iyf | 1 | -5.96 | [C@@H]1([C@@H](CC[C@H](O1)C)O)O | 9 |
| 2x6w_ligand_1_13.mol2 | 2x6w | 0.96875 | -6.56 | C[C@@H]1[C@H](CC[C@H](O1)O)O | 9 |
| 1m7d_ligand_1_6.mol2 | 1m7d | 0.96875 | -6.46 | O[C@H]1CC[C@H]([C@@H](O1)C)O | 9 |
| 2x85_ligand_1_13.mol2 | 2x85 | 0.96875 | -6.46 | C[C@@H]1[C@H](CC[C@@H](O1)O)O | 9 |
| 2x6y_ligand_1_13.mol2 | 2x6y | 0.96875 | -6.24 | [C@@H]1(CC[C@@H]([C@H](O1)C)O)O | 9 |
| 2iyf_ligand_1_3.mol2 | 2iyf | 0.96875 | -5.40 | [C@@H]1(C[C@@H]([C@H]([C@@H](O1)C)O)C)O | 10 |
100 ,
11

