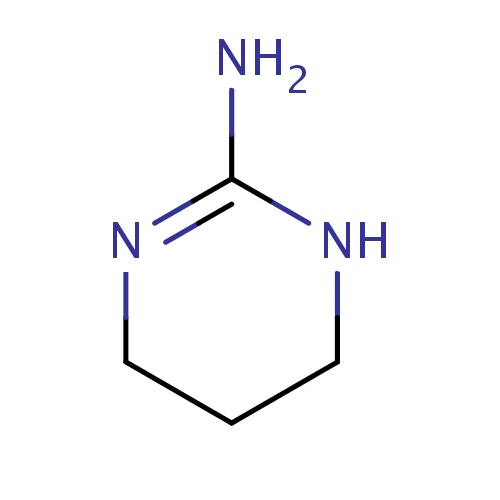
Common name
1,4,5,6-tetrahydropyrimidin-2-amine
IUPAC name
1,4,5,6-tetrahydropyrimidin-2-amine
SMILES
N1CCCN=C1N
Common name
1,4,5,6-tetrahydropyrimidin-2-amine
IUPAC name
1,4,5,6-tetrahydropyrimidin-2-amine
SMILES
N1CCCN=C1N
INCHI
InChI=1S/C4H9N3/c5-4-6-2-1-3-7-4/h1-3H2,(H3,5,6,7)
FORMULA
C4H9N3

Common name
1,4,5,6-tetrahydropyrimidin-2-amine
IUPAC name
1,4,5,6-tetrahydropyrimidin-2-amine
Molecular weight
99.134
clogP
0.761
clogS
-0.634
Frequency
0.0007
HBond Acceptor
1
HBond Donor
3
Total PolarSurface Area
50.41
Number of Rings
1
Rotatable Bond
0
| Drug ID | Common name | Structure CAS | Compound class | Therapeutic area |
|---|---|---|---|---|
| FDBD00200 | Capreomycin |
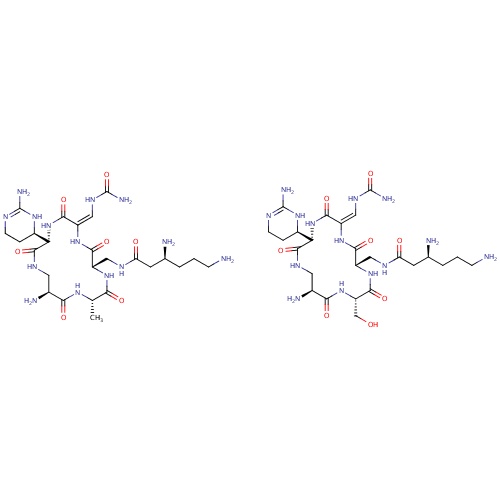
|
Anti-Bacterial Agents; Protein Synthesis Inhibitors; Antibiotics, Antitubercular; Antibiotics; Antimycobacterials; Antiinfectives for Systemic Use; Drugs for Treatment of Tuberculosis; | Used in the treatment of tuberculosis in combination with other drugs. |
| FDBD01613 | Enviomycin |
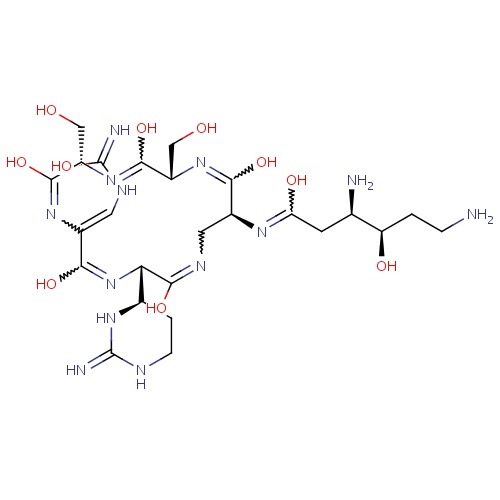
|
Anti-Bacterial Agents; Antibiotics, Antitubercular; |
2 ,
1
| FRAGNAME | PDBID | SIMILIRITY | XSCORE | SMILE | HAC |
|---|---|---|---|---|---|
| 1m21_ligand_frag_10.mol2 | 1m21 | 1 | -5.23 | C1NC(=NCC1)N | 7 |
| 1m21_ligand_1_10.mol2 | 1m21 | 0.914894 | -5.42 | C[C@H]1NC(=NCC1)N | 8 |
| 4s1g_ligand_1_0.mol2 | 4s1g | 0.68254 | -6.49 | CN1C(=O)C[C@@H](N=C1N)C | 10 |
| 4xx3_ligand_1_5.mol2 | 4xx3 | 0.68254 | -6.49 | CN1C(=O)C[C@@H]([NH]=C1N)C | 10 |
| 2va7_ligand_1_4.mol2 | 2va7 | 0.68254 | -6.48 | CC1(CC(=O)N(C(=N1)N)C)C | 11 |
| 4frs_ligand_frag_1.mol2 | 4frs | 0.68254 | -6.38 | C1C(=O)N(C(=N[C@H]1C)N)C | 10 |
| 4fs4_ligand_frag_3.mol2 | 4fs4 | 0.68254 | -6.23 | [C@@H]1(CC(=O)N(C(=N1)N)C)C | 10 |
| 2va7_ligand_frag_5.mol2 | 2va7 | 0.68254 | -6.19 | [C@@H]1(CC(=O)N(C(=N1)N)C)C | 10 |
100 ,
11

