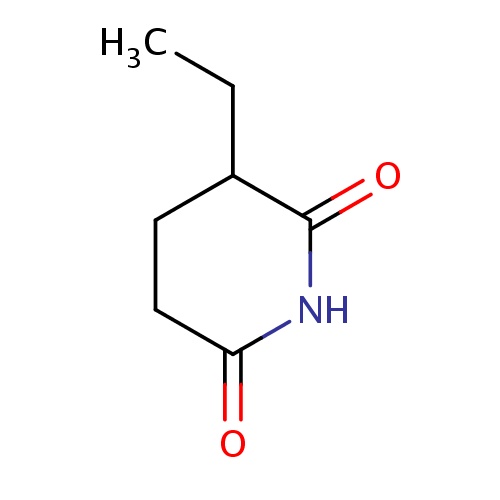
Common name
(3S)-3-ethylpiperidine-2,6-dione
IUPAC name
(3S)-3-ethylpiperidine-2,6-dione
SMILES
C(C)C1C(=O)NC(=O)CC1
Common name
(3S)-3-ethylpiperidine-2,6-dione
IUPAC name
(3S)-3-ethylpiperidine-2,6-dione
SMILES
C(C)C1C(=O)NC(=O)CC1
INCHI
InChI=1S/C7H11NO2/c1-2-5-3-4-6(9)8-7(5)10/h5H,2-4H2,1H3,(H,8,9,10)/t5-/m0/s1
FORMULA
C7H11NO2

Common name
(3S)-3-ethylpiperidine-2,6-dione
IUPAC name
(3S)-3-ethylpiperidine-2,6-dione
Molecular weight
141.168
clogP
1.206
clogS
-1.633
Frequency
0.0007
HBond Acceptor
2
HBond Donor
1
Total PolarSurface Area
46.17
Number of Rings
1
Rotatable Bond
1
| Drug ID | Common name | Structure CAS | Compound class | Therapeutic area |
|---|---|---|---|---|
| FDBD00241 | Aminoglutethimide |
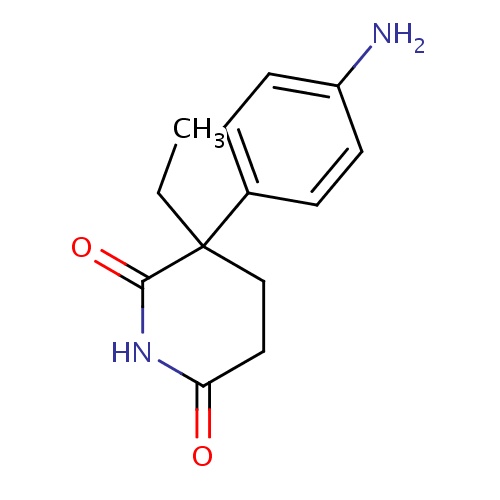
|
Antineoplastic Agents, Hormonal; Aromatase Inhibitors; Antineoplastic and Immunomodulating Agents; Endocrine Therapy; Hormone Antagonists and Related Agents; Cytochrome P-450 CYP1A2 Inhibitors; Cytochrome P-450 CYP1A2 Inducers; Cytochrome P-450 CYP2C19 Inducers; CYP3A4 Inhibitors; | For the suppression of adrenal function in selected patients with Cushing's syndrome, malignant neoplasm of the female breast, and carcinoma in situ of the breast. |
| FDBD01210 | Glutethimide |

|
Hypnotics and Sedatives; Nervous System; Psycholeptics; Piperidinedione Derivatives; CYP2D6 Inducers; CYP2D6 Inducers (strong); | For the treatment of insomnia. |
2 ,
1
| FRAGNAME | PDBID | SIMILIRITY | XSCORE | SMILE | HAC |
|---|---|---|---|---|---|
| 5bpe_ligand_2_51.mol2 | 5bpe | 0.914286 | -6.27 | C1C[C@H](C(=O)NC1)CC | 9 |
| 4n8e_ligand_1_2.mol2 | 4n8e | 0.888889 | -6.51 | C1CCC(CC1)N1C(=O)CCCC1 | 13 |
| 2ko7_ligand_2_0.mol2 | 2ko7 | 0.885714 | -5.81 | CC1CC(=O)N(C(=O)C1)C | 10 |
| 4tw7_ligand_2_121.mol2 | 4tw7 | 0.864865 | -8.36 | C1CCCCN1C(=O)CC1CCCCC1 | 15 |
| 3n7r_ligand_1_3.mol2 | 3n7r | 0.861111 | -6.28 | CN1C(=O)CCCCC1 | 9 |
| 3n7r_ligand_frag_1.mol2 | 3n7r | 0.861111 | -6.22 | C1CCCC(=O)NC1 | 8 |
| 1biw_ligand_2_9.mol2 | 1biw | 0.861111 | -6.11 | C1(=O)N(CCCCC1)CC | 10 |
| 1biw_ligand_1_0.mol2 | 1biw | 0.861111 | -5.91 | C1(=O)N(CCCCC1)C | 9 |
| 1mzc_ligand_frag_0.mol2 | 1mzc | 0.861111 | -5.90 | C1C(=O)N(CCCC1)C | 9 |
106 ,
11

