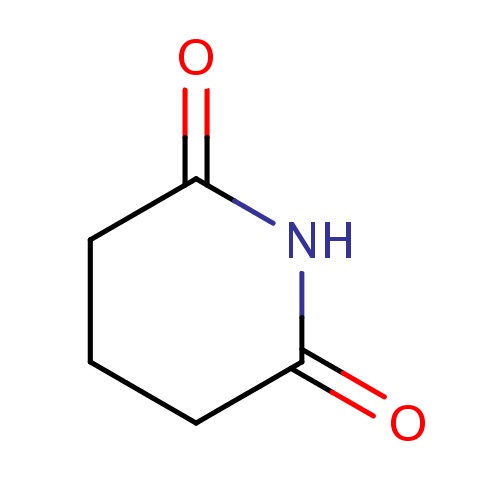
Common name
piperidine-2,6-dione
IUPAC name
piperidine-2,6-dione
SMILES
O=C1NC(=O)CCC1
Common name
piperidine-2,6-dione
IUPAC name
piperidine-2,6-dione
SMILES
O=C1NC(=O)CCC1
INCHI
InChI=1S/C5H7NO2/c7-4-2-1-3-5(8)6-4/h1-3H2,(H,6,7,8)
FORMULA
C5H7NO2

Common name
piperidine-2,6-dione
IUPAC name
piperidine-2,6-dione
Molecular weight
113.115
clogP
0.957
clogS
-1.039
Frequency
0.0021
HBond Acceptor
2
HBond Donor
1
Total PolarSurface Area
46.17
Number of Rings
1
Rotatable Bond
0
| Drug ID | Common name | Structure CAS | Compound class | Therapeutic area |
|---|---|---|---|---|
| FDBD00241 | Aminoglutethimide |
|
Antineoplastic Agents, Hormonal; Aromatase Inhibitors; Antineoplastic and Immunomodulating Agents; Endocrine Therapy; Hormone Antagonists and Related Agents; Cytochrome P-450 CYP1A2 Inhibitors; Cytochrome P-450 CYP1A2 Inducers; Cytochrome P-450 CYP2C19 Inducers; CYP3A4 Inhibitors; | For the suppression of adrenal function in selected patients with Cushing's syndrome, malignant neoplasm of the female breast, and carcinoma in situ of the breast. |
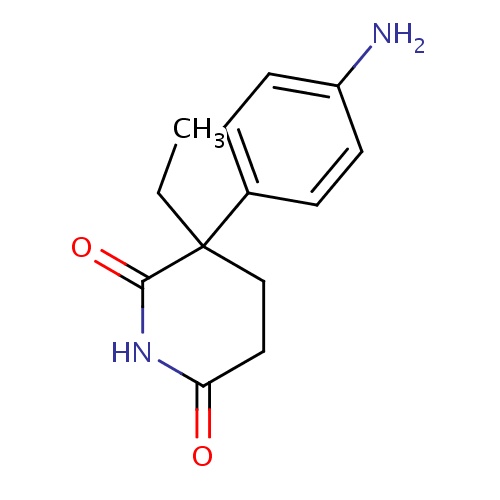
| FDBD00353 | Lenalidomide |

|
Antineoplastic Agents; Immunologic Factors; Immunosuppressive Agents; Angiogenesis Inhibitors; Antineoplastic and Immunomodulating Agents; | Lenalidomide is indicated for the treatment of multiple myeloma in combination with dexamethasone. It is also indicated for the treatment of patients with transfusion-dependent anemia due to low- or intermediate- risk myelodysplastic syndromes (MDS) associated with a deletion 5q cytogenetic abnormality with or without additional cytogenetic abnormalities. |
| FDBD00893 | Thalidomide |

|
Immunosuppressive Agents; Angiogenesis Inhibitors; Leprostatic Agents; Teratogens; Antineoplastic and Immunomodulating Agents; Cytochrome P-450 CYP1A2 Inhibitors; Cytochrome P-450 CYP2C9 Inhibitors; Cytochrome P-450 CYP1A2 Inducers; Cytochrome P-450 CYP2C9 Inducers; Cytochrome P-450 CYP2C19 Inducers; CYP2E1 Inhibitors; CYP2E1 Inducers; CYP2E1 Inducers (strong); | For the acute treatment of the cutaneous manifestations of moderate to severe erythema nodosum leprosum (ENL). Also for use as maintenance therapy for prevention and suppression of the cutaneous manifestations of ENL recurrence. |
| FDBD01210 | Glutethimide |

|
Hypnotics and Sedatives; Nervous System; Psycholeptics; Piperidinedione Derivatives; CYP2D6 Inducers; CYP2D6 Inducers (strong); | For the treatment of insomnia. |
| FDBD01571 | Pomalidomide |

|
Immunologic Factors; Immunosuppressive Agents; Angiogenesis Inhibitors; Antineoplastic and Immunomodulating Agents; Cytochrome P-450 CYP1A2 Inhibitors; Cytochrome P-450 CYP1A2 Inducers; CYP3A4 Inhibitors; | Pomalidomide is indicated for patients with multiple myeloma who have received at least two prior therapies including lenalidomide and bortezomib and have demonstrated disease progression on or within 60 days of completion of the last therapy. |
| FDBD02969 | cycloheximide |
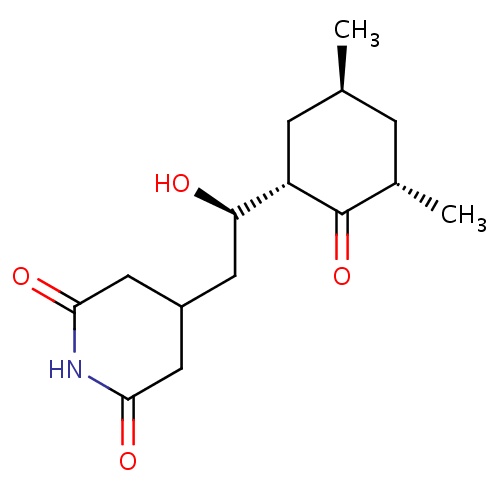
|
Fungicide | Fungicide |
6 ,
1
| FRAGNAME | PDBID | SIMILIRITY | XSCORE | SMILE | HAC |
|---|---|---|---|---|---|
| 4ci1_ligand_frag_0.mol2 | 4ci1 | 1 | -7.13 | O=C1NC(=O)CCC1 | 8 |
| 4ci3_ligand_frag_1.mol2 | 4ci3 | 1 | -7.12 | C1CCC(=O)NC1=O | 8 |
| 4ci2_ligand_frag_0.mol2 | 4ci2 | 1 | -7.10 | C1CCC(=O)NC1=O | 8 |
| 2ko7_ligand_frag_0.mol2 | 2ko7 | 1 | -5.51 | N1C(=O)CCCC1=O | 8 |
| 2ko7_ligand_1_1.mol2 | 2ko7 | 0.966667 | -5.72 | CC1CC(=O)NC(=O)C1 | 9 |
| 2ko7_ligand_1_0.mol2 | 2ko7 | 0.966667 | -5.60 | N1(C(=O)CCCC1=O)C | 9 |
| 2ko7_ligand_2_0.mol2 | 2ko7 | 0.935484 | -5.81 | CC1CC(=O)N(C(=O)C1)C | 10 |
| 2p16_ligand_frag_4.mol2 | 2p16 | 0.931034 | -6.07 | C1(=O)NCCCC1 | 7 |
| 4bti_ligand_frag_9.mol2 | 4bti | 0.931034 | -6.02 | N1CCCCC1=O | 7 |
| 4btu_ligand_frag_6.mol2 | 4btu | 0.931034 | -6.02 | N1CCCCC1=O | 7 |
123 ,
13

