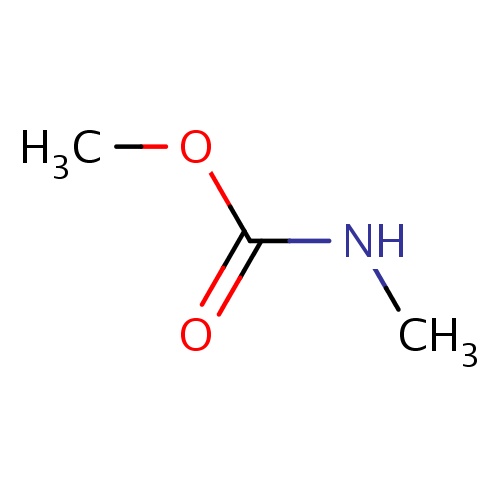
Common name
methyl N-methylcarbamate
IUPAC name
methyl N-methylcarbamate
SMILES
O(C(=O)NC)C
Common name
methyl N-methylcarbamate
IUPAC name
methyl N-methylcarbamate
SMILES
O(C(=O)NC)C
INCHI
InChI=1S/C3H7NO2/c1-4-3(5)6-2/h1-2H3,(H,4,5)
FORMULA
C3H7NO2

Common name
methyl N-methylcarbamate
IUPAC name
methyl N-methylcarbamate
Molecular weight
89.093
clogP
-0.655
clogS
-0.296
Frequency
0.0048
HBond Acceptor
2
HBond Donor
1
Total PolarSurface Area
38.33
Number of Rings
0
Rotatable Bond
1
| Drug ID | Common name | Structure CAS | Compound class | Therapeutic area |
|---|---|---|---|---|
| FDBD00376 | Ritonavir |

|
Protease Inhibitors; HIV Protease Inhibitors; Antiinfectives for Systemic Use; Direct Acting Antivirals; Antivirals for Systemic Use; Cytochrome P-450 CYP1A2 Inhibitors; Cytochrome P-450 CYP2C9 Inhibitors; Cytochrome P-450 CYP1A2 Inducers; Cytochrome P-450 CYP2C8 Inhibitors; Cytochrome P-450 CYP3A Inhibitors; Cytochrome P-450 CYP2C9 Inducers; Cytochrome P-450 CYP2C19 Inducers; Cytochrome P-450 CYP2C8 Inducers; Cytochrome P-450 CYP2B6 Inducers; Cytochrome P-450 CYP2B6 Inhibitors; CYP2B6 Inhibitors (strong); CYP2D6 Inducers; CYP2D6 Inducers (strong); CYP2E1 Inhibitors; CYP2E1 Inducers; CYP2E1 Inducers (strong); CYP3A4 Inhibitors; Combined Inducers of CYP3A4 and P-glycoprotein; Combined Inhibitors of CYP3A4 and P-glycoprotein; | Indicated in combination with other antiretroviral agents for the treatment of HIV-infection. |
| FDBD00923 | Atazanavir |
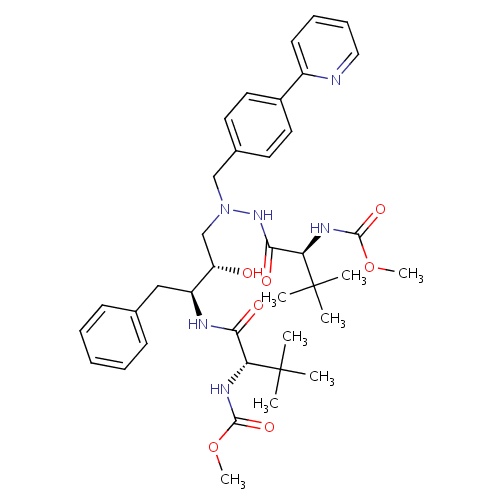
|
Anti-HIV Agents; Protease Inhibitors; HIV Protease Inhibitors; Antiinfectives for Systemic Use; Direct Acting Antivirals; Antivirals for Systemic Use; Cytochrome P-450 CYP2C9 Inhibitors; Cytochrome P-450 CYP2C9 Inducers; CYP3A4 Inhibitors; Combined Inhibitors of CYP3A4 and P-glycoprotein; | Used in combination with other antiretroviral agents for the treatment of HIV-1 infection, as well as postexposure prophylaxis of HIV infection in individuals who have had occupational or nonoccupational exposure to potentially infectious body fluids of a person known to be infected with HIV when that exposure represents a substantial risk for HIV transmission. |
| FDBD01581 | Riociguat |

|
Riociguat is indicated for the treatment of adults with persistent/recurrent chronic thromboembolic pulmonary hypertension (CTEPH), (WHO Group 4) after surgical treatment, or inoperable CTEPH, to improve exercise capacity and WHO functional class. | |
| FDBD01634 | Ledipasvir |

|
Antiviral Agents; Antiinfectives for Systemic Use; Direct Acting Antivirals; Antivirals for Systemic Use; | Ledipasvir in combination with sofosbuvir, or in combination with sofosbuvir and ribavirin, is indicated for the treatment of chronic hepatitis C (CHC) genotype 1 infection in adults. |
| FDBD01652 | Cobicistat |
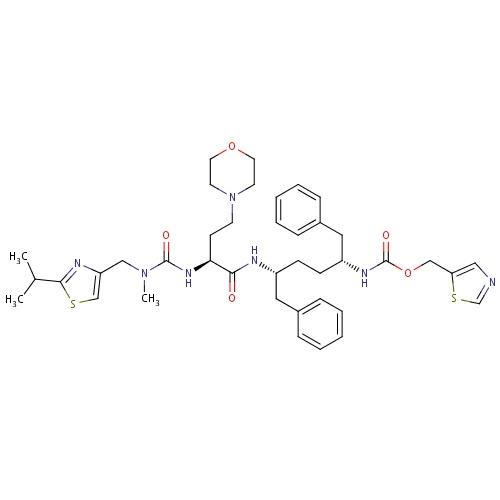
|
Anti-HIV Agents; Antiinfectives for Systemic Use; Direct Acting Antivirals; Antivirals for Systemic Use; Cytochrome P-450 CYP3A Inhibitors; CYP2D6 Inducers; CYP2D6 Inducers (strong); | Cobicistat is a CYP3A inhibitor indicated to increase systemic exposure of atazanavir or darunavir (once daily dosing regimen) in combination with other antiretroviral agents in the treatment of HIV-1 infection. It is not interchangeable with ritonavir to increase systemic exposure of darunavir 600 mg twice daily, fosamprenavir, saquinavir, or tipranavir due to lack of exposure data. The use of cobicistat is not recommended with darunavir 600 mg twice daily, fosamprenavir, saquinavir or tipranavir. Complex or unknown mechanisms of drug interactions preclude extrapolation of ritonavir drug interactions to certain cobicistat interactions. Cobicistat and ritonavir when administered with either atazanavir or darunavir may result in different drug interactions when used with concomitant medications. |
| FDBD01682 | Daclatasvir |

|
Antiinfectives for Systemic Use; Direct Acting Antivirals; Antivirals for Systemic Use; CYP3A4 Inhibitors; | Daklinza is used to treat patients who have chronic hepatitis C virus (HCV) genotype 3 infection. Daklinza is typically taken in conjunction with sofosbuvir. (2). |
| FDBD01793 | Ombitasvir |
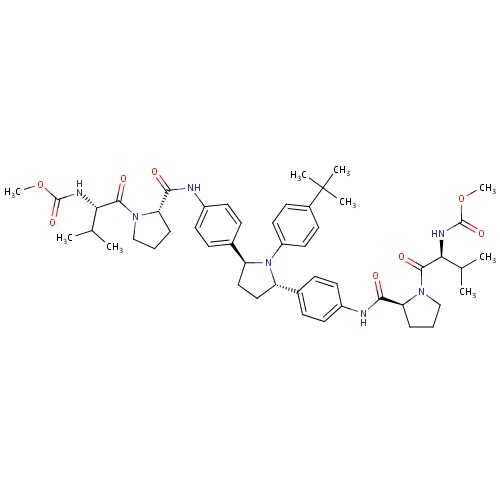
|
Antiinfectives for Systemic Use; Direct Acting Antivirals; Antivirals for Systemic Use; | For use in combination with paritaprevir, ritonavir and dasabuvir for the treatment of HCV genotype 1, and with paritaprevir and ritonavir for the treatment of HCV genotype 4. |
| FDBD01836 | Elbasvir |
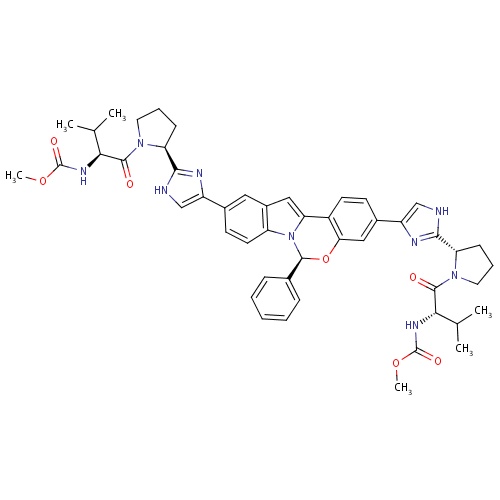
|
; | |
| FDBD01870 | furathiocarb |

|
Insecticide | Insecticide |
| FDBD02481 | dichlormate |

|
Herbicide | Herbicide |
14 ,
2
| FRAGNAME | PDBID | SIMILIRITY | XSCORE | SMILE | HAC |
|---|---|---|---|---|---|
| 3eql_ligand_frag_1.mol2 | 3eql | 1 | -5.58 | N(C)C(=O)OC | 6 |
| 4tmn_ligand_2_41.mol2 | 4tmn | 1 | -5.54 | COC(=O)NC | 6 |
| 4imq_ligand_2_1.mol2 | 4imq | 1 | -5.49 | CNC(=O)OC | 6 |
| 2pj1_ligand_2_8.mol2 | 2pj1 | 1 | -5.44 | COC(=O)NC | 6 |
| 1tu6_ligand_2_25.mol2 | 1tu6 | 1 | -5.43 | CNC(=O)OC | 6 |
129 ,
13

