
IUPAC name
(2S)-2-{[hydroxy(methoxy)methylidene]amino}-1-[(2S)-2-[5-(4'-{2-[(2S)-1-[(2S)-2-{[hydroxy(methoxy)methylidene]amino}-3-methylbutanoyl]pyrrolidin-2-yl]-1H-imidazol-5-yl}-[1,1'-biphenyl]-4-yl)-1H-imidazol-2-yl]pyrrolidin-1-yl]-3-methylbutan-1-one dihydrochloride
SMILES
Cl.Cl.[H][C@](N=C(O)OC)(C(C)C)C(=O)N1CCC[C@@]1([H])C1=NC=C(N1)C1=CC=C(C=C1)C1=CC=C(C=C1)C1=CN=C(N1)[C@]1([H])CCCN1C(=O)[C@@]([H])(N=C(O)OC)C(C)C
Compound class
Antiinfectives for Systemic Use; Direct Acting Antivirals; Antivirals for Systemic Use; CYP3A4 Inhibitors;
Therapeutic area
Daklinza is used to treat patients who have chronic hepatitis C virus (HCV) genotype 3 infection. Daklinza is typically taken in conjunction with sofosbuvir. (2).
Common name
Daclatasvir
IUPAC name
(2S)-2-{[hydroxy(methoxy)methylidene]amino}-1-[(2S)-2-[5-(4'-{2-[(2S)-1-[(2S)-2-{[hydroxy(methoxy)methylidene]amino}-3-methylbutanoyl]pyrrolidin-2-yl]-1H-imidazol-5-yl}-[1,1'-biphenyl]-4-yl)-1H-imidazol-2-yl]pyrrolidin-1-yl]-3-methylbutan-1-one dihydrochloride
SMILES
Cl.Cl.[H][C@](N=C(O)OC)(C(C)C)C(=O)N1CCC[C@@]1([H])C1=NC=C(N1)C1=CC=C(C=C1)C1=CC=C(C=C1)C1=CN=C(N1)[C@]1([H])CCCN1C(=O)[C@@]([H])(N=C(O)OC)C(C)C
INCHI
InChI=1S/C40H50N8O6.2ClH/c1-23(2)33(45-39(51)53-5)37(49)47-19-7-9-31(47)35-41-21-29(43-35)27-15-11-25(12-16-27)26-13-17-28(18-14-26)30-22-42-36(44-30)32-10-8-20-48(32)38(50)34(24(3)4)46-40(52)54-6;;/h11-18,21-24,31-34H,7-10,19-20H2,1-6H3,(H,41,43)(H,42,44)(H,45,51)(H,46,52);2*1H/t31-,32-,33-,34-;;/m0../s1
FORMULA
C40H52Cl2N8O6

Common name
Daclatasvir
IUPAC name
(2S)-2-{[hydroxy(methoxy)methylidene]amino}-1-[(2S)-2-[5-(4'-{2-[(2S)-1-[(2S)-2-{[hydroxy(methoxy)methylidene]amino}-3-methylbutanoyl]pyrrolidin-2-yl]-1H-imidazol-5-yl}-[1,1'-biphenyl]-4-yl)-1H-imidazol-2-yl]pyrrolidin-1-yl]-3-methylbutan-1-one dihydrochloride
Molecular weight
738.875
clogP
6.179
clogS
-6.970
HBond Acceptor
10
HBond Donor
4
Total Polar Surface Area
181.62
Number of Rings
6
Rotatable Bond
15
| Drug ID | Common name | Structure CAS | SMILE | Frequency |
|---|---|---|---|---|
| FDBF00002 | pyrrolidine-1-carbaldehyde |
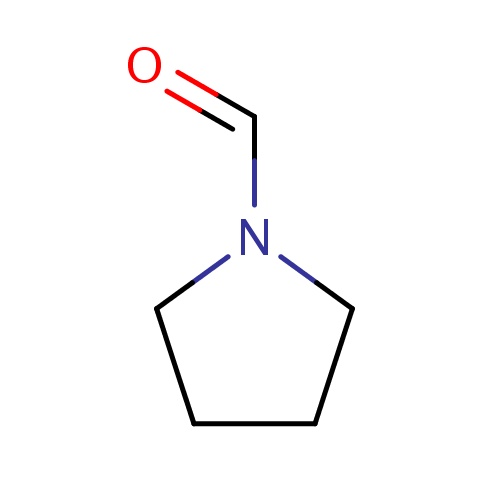
|
C(=O)N1CCCC1 | 0.0041 |
| FDBF00003 | formamide |
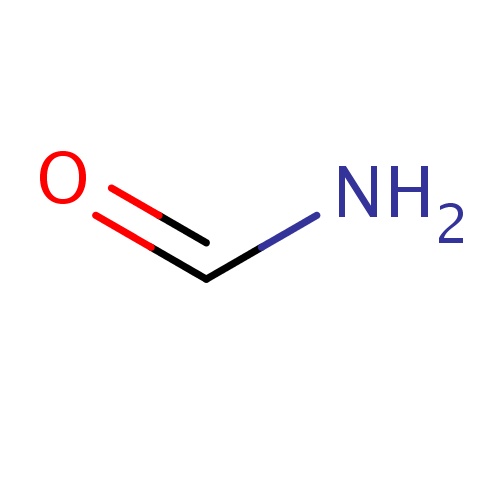
|
C(=O)N | 0.1240 |
| FDBF00011 | imidazole |
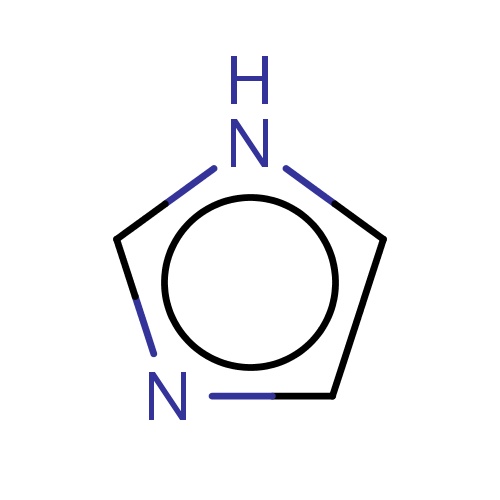
|
[nH]1ccnc1 | 0.0175 |
| FDBF00012 | isobutane |
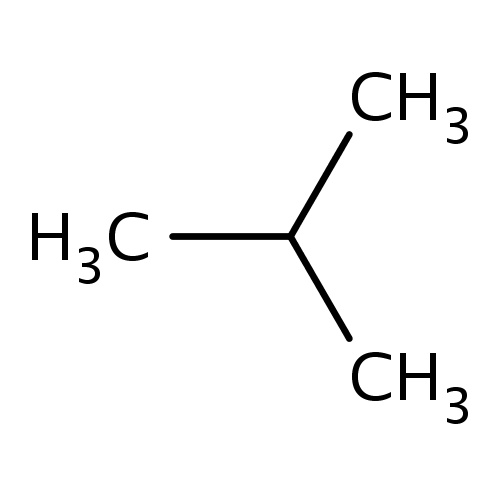
|
C(C)(C)C | 0.0611 |
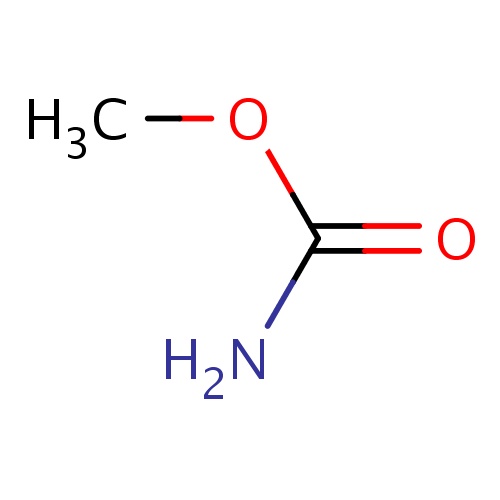
| FDBF00692 | methyl carbamate |
|
O(C(=O)N)C | 0.0117 |
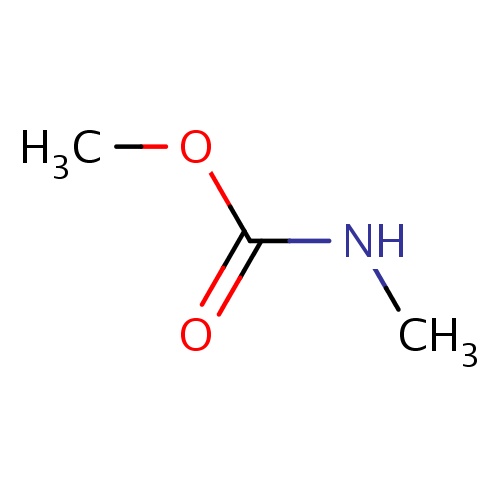
| FDBF01131 | methyl N-methylcarbamate |
|
O(C(=O)NC)C | 0.0048 |
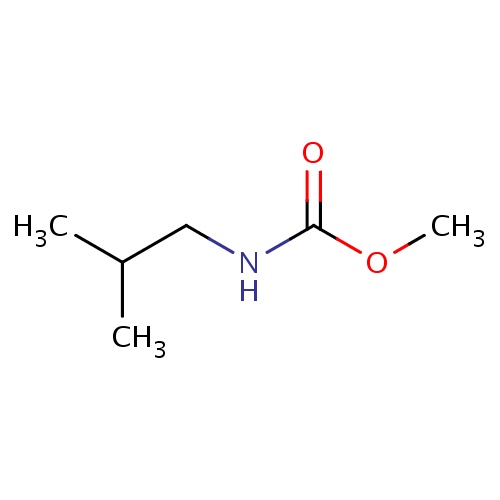
| FDBF03989 | methyl N-isobutylcarbamate |
|
CC(C)CNC(=O)OC | 0.0017 |
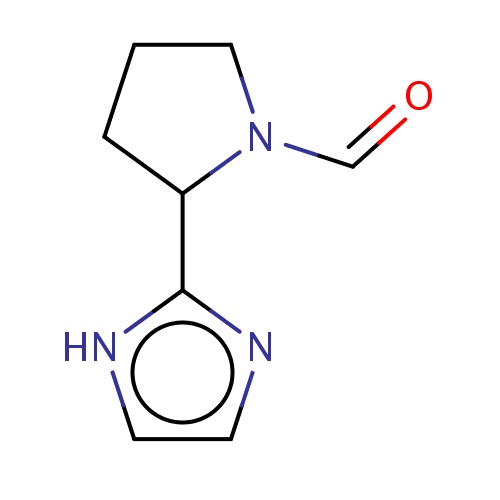
| FDBF04190 | (2S)-2-(1H-imidazol-2-yl)pyrrolidine-1-carbaldehyde |
|
c1c[nH]c(n1)C2CCCN2C=O | 0.0007 |
| FDBF04193 | 3-methyl-1-pyrrolidin-1-yl-butan-1-one |
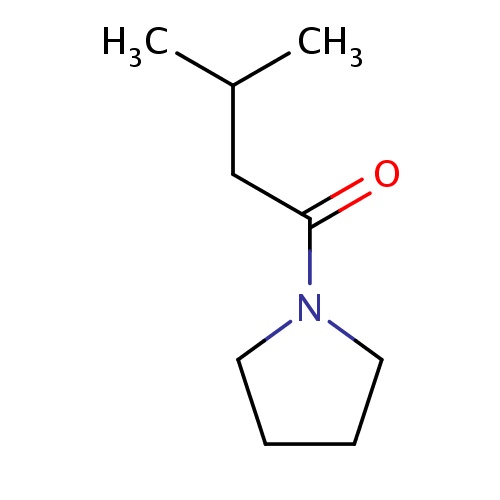
|
C(C(C)C)C(=O)N1CCCC1 | 0.0010 |
| FDBF04195 | 1-[(2S)-2-(5-phenyl-1H-imidazol-2-yl)pyrrolidin-1-yl]ethanone |
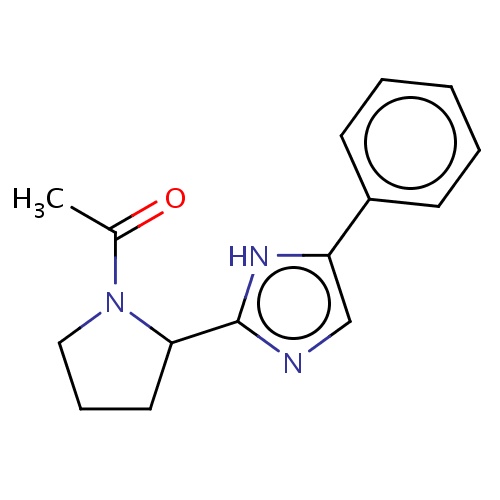
|
CC(=O)N1C(CCC1)c2[nH]c(cn2)c3ccccc3 | 0.0003 |

