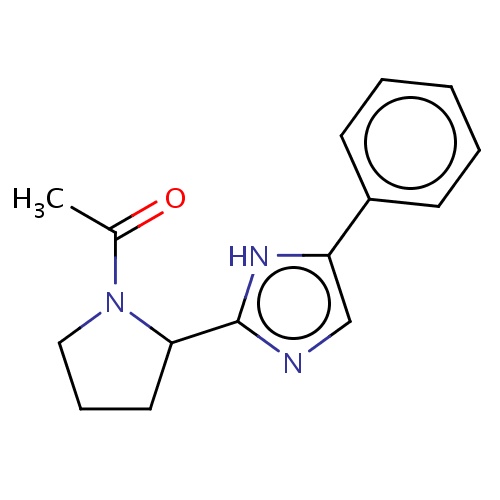
Common name
1-[(2S)-2-(5-phenyl-1H-imidazol-2-yl)pyrrolidin-1-yl]ethanone
IUPAC name
1-[(2S)-2-(5-phenyl-1H-imidazol-2-yl)pyrrolidin-1-yl]ethanone
SMILES
CC(=O)N1C(CCC1)c2[nH]c(cn2)c3ccccc3
Common name
1-[(2S)-2-(5-phenyl-1H-imidazol-2-yl)pyrrolidin-1-yl]ethanone
IUPAC name
1-[(2S)-2-(5-phenyl-1H-imidazol-2-yl)pyrrolidin-1-yl]ethanone
SMILES
CC(=O)N1C(CCC1)c2[nH]c(cn2)c3ccccc3
INCHI
InChI=1S/C15H17N3O/c1-11(19)18-9-5-8-14(18)15-16-10-13(17-15)12-6-3-2-4-7-12/h2-4,6-7,10,14H,5,8-9H2,1H3,(H,16,17)/t14-/m0/s1
FORMULA
C15H17N3O

Common name
1-[(2S)-2-(5-phenyl-1H-imidazol-2-yl)pyrrolidin-1-yl]ethanone
IUPAC name
1-[(2S)-2-(5-phenyl-1H-imidazol-2-yl)pyrrolidin-1-yl]ethanone
Molecular weight
255.315
clogP
2.806
clogS
-3.399
Frequency
0.0003
HBond Acceptor
2
HBond Donor
1
Total PolarSurface Area
48.99
Number of Rings
3
Rotatable Bond
3
| Drug ID | Common name | Structure CAS | Compound class | Therapeutic area |
|---|---|---|---|---|
| FDBD01682 | Daclatasvir |

|
Antiinfectives for Systemic Use; Direct Acting Antivirals; Antivirals for Systemic Use; CYP3A4 Inhibitors; | Daklinza is used to treat patients who have chronic hepatitis C virus (HCV) genotype 3 infection. Daklinza is typically taken in conjunction with sofosbuvir. (2). |
| FRAGNAME | PDBID | SIMILIRITY | XSCORE | SMILE | HAC |
|---|---|---|---|---|---|
| 4ty6_ligand_4_56.mol2 | 4ty6 | 0.771429 | -6.86 | c1cc(ccc1)c1[nH+]c([C@H](C)NC=O)[nH]c1 | 16 |
| 4ty6_ligand.mol2 | 4ty6 | 0.70922 | -9.64 | C1[C@@H](C(=O)N[C@H](c2nc(c[nH]2)c2ccc(cc2)C(=O)N)Cc2ccccc2)CC[C@@H](C1)C[NH3+] | 34 |
| 4ty6_ligand_3_38.mol2 | 4ty6 | 0.704762 | -6.62 | c1cc(ccc1)c1[nH+]c(CNC=O)[nH]c1 | 15 |
| 4x6o_ligand_3_92.mol2 | 4x6o | 0.647059 | -7.00 | c1(ccccc1)c1[nH+]c([nH]c1Cl)CNC=O | 16 |
| 4crd_ligand_3_17.mol2 | 4crd | 0.647059 | -6.86 | c1ccccc1c1c([nH+]c([nH]1)CNC=O)Cl | 16 |
| 1ouk_ligand_2_18.mol2 | 1ouk | 0.639344 | -7.54 | c1c([nH]c([n+]1C)C1CC[NH2+]CC1)c1ccccc1 | 18 |
| 3cd0_ligand_2_7.mol2 | 3cd0 | 0.632479 | -6.58 | [nH+]1c(c[nH]c1C(=O)NC)c1ccc(cc1)F | 16 |
| 3cd0_ligand_3_6.mol2 | 3cd0 | 0.604478 | -7.38 | [nH]1c(c[n+](c1C(=O)NC)C(C)C)c1ccc(cc1)F | 19 |
| 3cd0_ligand_4_11.mol2 | 3cd0 | 0.582734 | -7.51 | Cc1c([nH]c([n+]1C(C)C)C(=O)NC)c1ccc(cc1)F | 20 |
| 3k3i_ligand.mol2 | 3k3i | 0.578947 | -9.95 | [NH2+]1[C@H](c2nc(c3ccc(Br)cc3)c[nH]2)Cc2c(cccc2)C1 | 23 |

