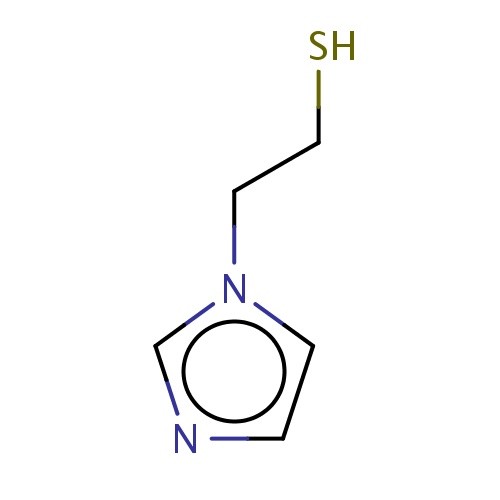
Common name
2-imidazol-1-ylethanethiol
IUPAC name
2-imidazol-1-ylethanethiol
SMILES
C(Cn1ccnc1)S
Common name
2-imidazol-1-ylethanethiol
IUPAC name
2-imidazol-1-ylethanethiol
SMILES
C(Cn1ccnc1)S
INCHI
InChI=1S/C5H8N2S/c8-4-3-7-2-1-6-5-7/h1-2,5,8H,3-4H2
FORMULA
C5H8N2S

Common name
2-imidazol-1-ylethanethiol
IUPAC name
2-imidazol-1-ylethanethiol
Molecular weight
129.203
clogP
-0.327
clogS
-0.841
Frequency
0.0007
HBond Acceptor
0
HBond Donor
2
Total PolarSurface Area
58.47
Number of Rings
1
Rotatable Bond
2
| Drug ID | Common name | Structure CAS | Compound class | Therapeutic area |
|---|---|---|---|---|
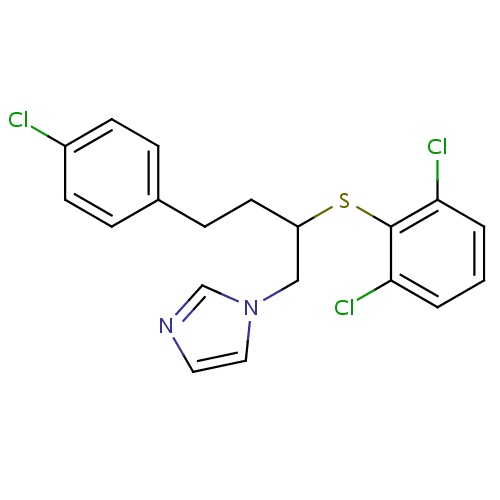
| FDBD00504 | Butoconazole |
|
Antifungal Agents; Genito Urinary System and Sex Hormones; Gynecological Antiinfectives and Antiseptics; Imidazole Derivatives; | For the local treatment of vulvovaginal candidiasis (infections caused by Candida). |
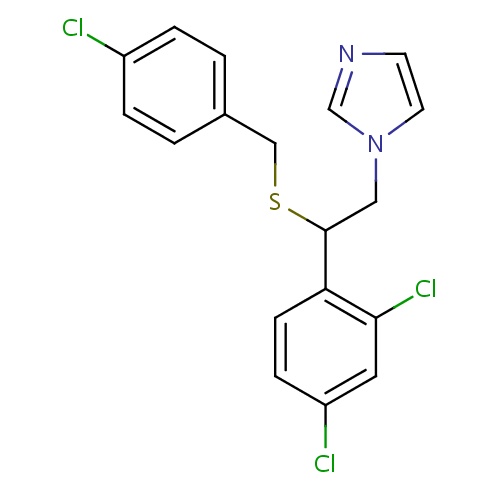
| FDBD01504 | Sulconazole |
|
Dermatologicals; Imidazole and Triazole Derivatives; Antifungals for Topical Use; Antifungals for Dermatological Use; Cytochrome P-450 CYP1A2 Inhibitors; Cytochrome P-450 CYP2C9 Inhibitors; Cytochrome P-450 CYP1A2 Inducers; Cytochrome P-450 CYP2C9 Inducers; | Sulconazole solution 1.0% is indicated for the treatment of tinea cruris and tinea corporis caused by Trichophyton rubrum, Trichophyton mentagrophytes, Epidermophyton floccosum, and Microsporum canis; and for the treatment of tinea versicolor. Effectiveness has not been proven in tinea pedis (athlete's foot). |
2 ,
1
| FRAGNAME | PDBID | SIMILIRITY | XSCORE | SMILE | HAC |
|---|---|---|---|---|---|
| 4fbe_ligand_2_8.mol2 | 4fbe | 0.727273 | -6.23 | c1c[n+](c[nH]1)CC | 7 |
| 4f9u_ligand_2_8.mol2 | 4f9u | 0.727273 | -6.18 | c1c[n+](c[nH]1)CC | 7 |
| 4mhy_ligand_2_8.mol2 | 4mhy | 0.727273 | -6.18 | c1c[n+](c[nH]1)CC | 7 |
| 4fai_ligand_2_7.mol2 | 4fai | 0.727273 | -6.17 | C([n+]1cc[nH]c1)C | 7 |
| 4f9v_ligand_2_7.mol2 | 4f9v | 0.727273 | -6.11 | C(C)[n+]1cc[nH]c1 | 7 |
| 4mhz_ligand_2_8.mol2 | 4mhz | 0.727273 | -6.08 | C(C)[n+]1cc[nH]c1 | 7 |
| 5dtj_ligand_2_20.mol2 | 5dtj | 0.727273 | -6.01 | CC[n+]1cc[nH]c1 | 7 |
| 5afx_ligand_2_45.mol2 | 5afx | 0.727273 | -5.90 | CC[n+]1cc[nH]c1 | 7 |
148 ,
15

