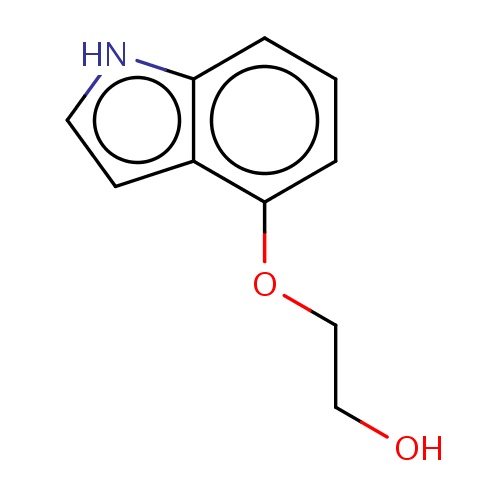
Common name
2-(1H-indol-4-yloxy)ethanol
IUPAC name
2-(1H-indol-4-yloxy)ethanol
SMILES
C(CO)Oc1c2c([nH]cc2)ccc1
Common name
2-(1H-indol-4-yloxy)ethanol
IUPAC name
2-(1H-indol-4-yloxy)ethanol
SMILES
C(CO)Oc1c2c([nH]cc2)ccc1
INCHI
InChI=1S/C10H11NO2/c12-6-7-13-10-3-1-2-9-8(10)4-5-11-9/h1-5,11-12H,6-7H2
FORMULA
C10H11NO2

Common name
2-(1H-indol-4-yloxy)ethanol
IUPAC name
2-(1H-indol-4-yloxy)ethanol
Molecular weight
177.200
clogP
2.206
clogS
-2.499
Frequency
0.0003
HBond Acceptor
2
HBond Donor
2
Total PolarSurface Area
45.25
Number of Rings
2
Rotatable Bond
3
| Drug ID | Common name | Structure CAS | Compound class | Therapeutic area |
|---|---|---|---|---|
| FDBD00815 | Pindolol |
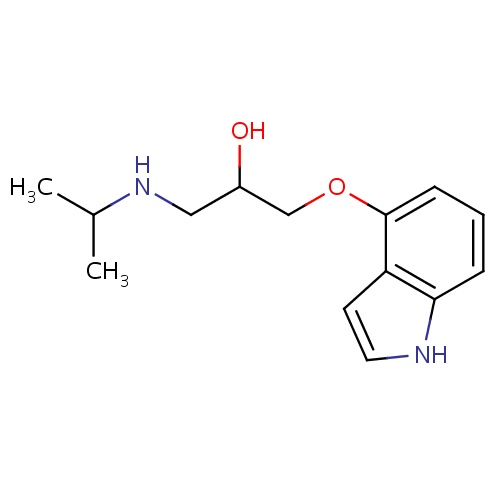
|
Antihypertensive Agents; Vasodilator Agents; Serotonin Antagonists; Adrenergic beta-Antagonists; Cardiovascular System; Beta Blocking Agents; Beta Blocking Agents, Non-Selective; CYP2D6 Inducers; CYP2D6 Inducers (strong); | For the management of hypertension, edema, ventricular tachycardias, and atrial fibrillation. |
1 ,
1
| FRAGNAME | PDBID | SIMILIRITY | XSCORE | SMILE | HAC |
|---|---|---|---|---|---|
| 4gmc_ligand_1_1.mol2 | 4gmc | 0.884615 | -6.85 | C[N@H+]1[C@@H]2CCCC[C@@H]2C[C@@H]1[C@H]1CCOC1 | 15 |
| 4gmc_ligand_frag_0.mol2 | 4gmc | 0.884615 | -6.80 | C1C[C@@H]2[C@H](CC1)C[C@@H]([NH2+]2)[C@H]1CCOC1 | 14 |
| 2iog_ligand_4_321.mol2 | 2iog | 0.862745 | -7.25 | C(O[C@@H]1C[C@@H]2[NH2+]C[C@H](C)[C@H]2CC1)C | 13 |
| 2iog_ligand_3_161.mol2 | 2iog | 0.862745 | -6.97 | C(O[C@@H]1C[C@@H]2[NH2+]CC[C@H]2CC1)C | 12 |
| 4fn5_ligand_2_10.mol2 | 4fn5 | 0.843137 | -6.51 | C[C@@H]1C[NH2+][C@H]2[C@H]1[C@H](CCC2)OC | 12 |
| 4fn5_ligand_1_2.mol2 | 4fn5 | 0.843137 | -6.21 | C1C[C@H]2[C@@H](CCC[C@@H]2OC)[NH2+]1 | 11 |
| 1qpf_ligand_3_80.mol2 | 1qpf | 0.833333 | -5.51 | C1[C@@H](C[C@H]2[C@@H](C1)[NH2+]CC2)O[C@@H]1CCCC[C@H]1OC | 18 |
| 1qpl_ligand_3_55.mol2 | 1qpl | 0.833333 | -5.47 | C1C[C@H]([C@@H](CC1)O[C@H]1CC[C@@H]2[C@H](C1)CC[N@@H+]2C)OC | 19 |
| 1qb9_ligand_3_6.mol2 | 1qb9 | 0.830189 | -6.90 | C1(CC[NH2+]CC1)O[C@@H]1CC[C@H]2[C@H]([N@@H+](C)[C@@H]3[C@@H]2CCCC3)C1 | 21 |
100 ,
11

