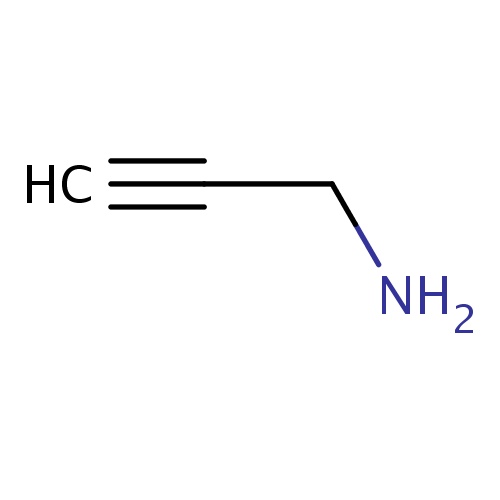
Common name
prop-2-yn-1-amine
IUPAC name
prop-2-yn-1-amine
SMILES
NCC#C
Common name
prop-2-yn-1-amine
IUPAC name
prop-2-yn-1-amine
SMILES
NCC#C
INCHI
InChI=1S/C3H5N/c1-2-3-4/h1H,3-4H2
FORMULA
C3H5N

Common name
prop-2-yn-1-amine
IUPAC name
prop-2-yn-1-amine
Molecular weight
55.079
clogP
-0.372
clogS
0.183
Frequency
0.0007
HBond Acceptor
0
HBond Donor
2
Total PolarSurface Area
26.02
Number of Rings
0
Rotatable Bond
1
| Drug ID | Common name | Structure CAS | Compound class | Therapeutic area |
|---|---|---|---|---|
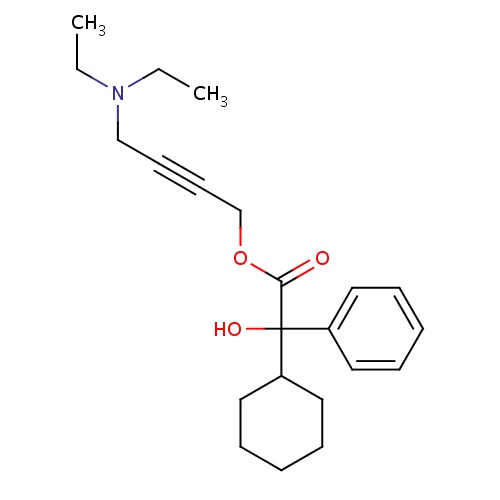
| FDBD00913 | Oxybutynin |
|
Muscarinic Antagonists; Parasympatholytics; Cholinergic Antagonists; Antispasmodics; Muscle Relaxants, Genitourinary; Genito Urinary System and Sex Hormones; Drugs for Urinary Frequency and Incontinence; Urological Agents; Cytochrome P-450 CYP2C8 Inhibitors; Cytochrome P-450 CYP2C8 Inducers; CYP2D6 Inducers; CYP2D6 Inducers (strong); CYP3A4 Inhibitors; | For the treatment of overactive bladder. |
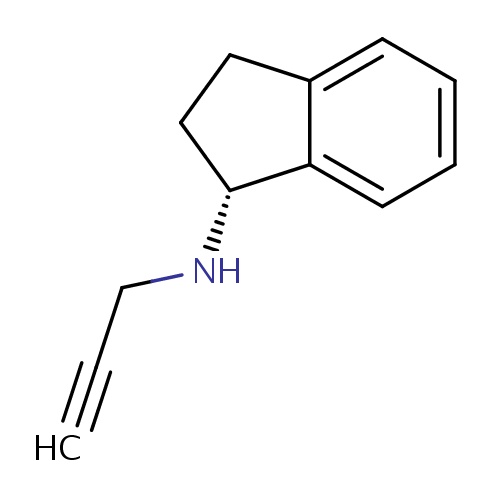
| FDBD01163 | Rasagiline |
|
Neuroprotective Agents; Monoamine Oxidase Inhibitors; Nervous System; Anti-Parkinson Drugs; Dopaminergic Agents; Monoamine Oxidase B Inhibitors; Cytochrome P-450 CYP1A2 Inhibitors; Cytochrome P-450 CYP1A2 Inducers; | For the treatment of the signs and symptoms of idiopathic Parkinsons disease as initial monotherapy and as adjunct therapy to levodopa. |
2 ,
1
| FRAGNAME | PDBID | SIMILIRITY | XSCORE | SMILE | HAC |
|---|---|---|---|---|---|
| 2a8h_ligand_1_5.mol2 | 2a8h | 1 | -5.41 | C([NH3+])C#C | 4 |
| 1ci7_ligand_2_25.mol2 | 1ci7 | 1 | -4.95 | C(C#C)[NH3+] | 4 |
| 2a8h_ligand_2_14.mol2 | 2a8h | 0.75 | -5.89 | CC#CC[NH3+] | 5 |
| 2pjt_ligand_2_0.mol2 | 2pjt | 0.75 | -5.76 | CC#CC[NH3+] | 5 |
| 4qvx_ligand_2_13.mol2 | 4qvx | 0.666667 | -5.05 | C(C#C)[NH+](C)C | 6 |
| 1ci7_ligand_3_38.mol2 | 1ci7 | 0.666667 | -4.95 | C(C#C)[NH2+]C | 5 |
| 1a99_ligand_1_2.mol2 | 1a99 | 0.5 | -5.92 | CC[NH3+] | 3 |
| 1pot_ligand_1_5.mol2 | 1pot | 0.5 | -5.90 | C([NH3+])C | 3 |
1220 ,
123

