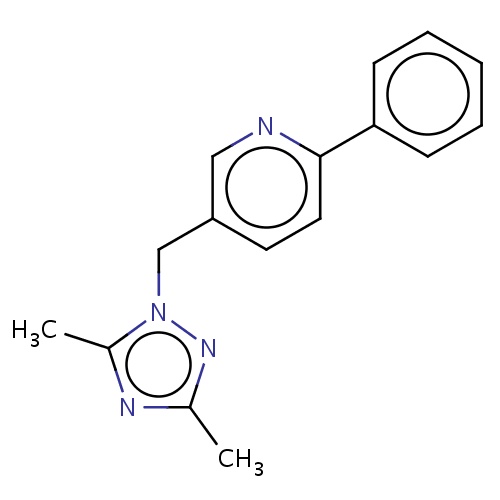
Common name
5-[(3,5-dimethyl-1,2,4-triazol-1-yl)methyl]-2-phenyl-pyridine
IUPAC name
5-[(3,5-dimethyl-1,2,4-triazol-1-yl)methyl]-2-phenyl-pyridine
SMILES
n1(nc(nc1C)C)Cc2cnc(cc2)c3ccccc3
Common name
5-[(3,5-dimethyl-1,2,4-triazol-1-yl)methyl]-2-phenyl-pyridine
IUPAC name
5-[(3,5-dimethyl-1,2,4-triazol-1-yl)methyl]-2-phenyl-pyridine
SMILES
n1(nc(nc1C)C)Cc2cnc(cc2)c3ccccc3
INCHI
InChI=1S/C16H16N4/c1-12-18-13(2)20(19-12)11-14-8-9-16(17-10-14)15-6-4-3-5-7-15/h3-10H,11H2,1-2H3
FORMULA
C16H16N4

Common name
5-[(3,5-dimethyl-1,2,4-triazol-1-yl)methyl]-2-phenyl-pyridine
IUPAC name
5-[(3,5-dimethyl-1,2,4-triazol-1-yl)methyl]-2-phenyl-pyridine
Molecular weight
265.333
clogP
2.130
clogS
-4.673
Frequency
0.0003
HBond Acceptor
2
HBond Donor
1
Total PolarSurface Area
45.45
Number of Rings
3
Rotatable Bond
3
| Drug ID | Common name | Structure CAS | Compound class | Therapeutic area |
|---|---|---|---|---|
| FDBD01145 | Forasartan |
|
Angiotensin Receptor Antagonists; Angiotensin II Receptor Antagonists; | For the treatment of hypertension. |
1 ,
1
| FRAGNAME | PDBID | SIMILIRITY | XSCORE | SMILE | HAC |
|---|---|---|---|---|---|
| 5aix_ligand_2_5.mol2 | 5aix | 0.528926 | -7.96 | c1cccc(c1)c1ccc(C(=O)N)cn1 | 15 |
| 3iph_ligand_2_1.mol2 | 3iph | 0.503937 | -8.33 | c1(ccccc1C)c1ccc(cn1)C(=O)N | 16 |
| 2c5y_ligand_3_0.mol2 | 2c5y | 0.496296 | -7.89 | c1(ncncc1)c1cc(ccc1)C[n+]1c[nH]cn1 | 18 |
| 2c5x_ligand_3_0.mol2 | 2c5x | 0.496296 | -7.75 | c1(cc(ccc1)c1ncncc1)C[n+]1c[nH]cn1 | 18 |
| 4ejn_ligand_3_19.mol2 | 4ejn | 0.47619 | -8.78 | Cc1ccc(cc1)[n+]1c2c(ccc(c3ccccc3)n2)[nH]c1 | 22 |
| 5aix_ligand_4_10.mol2 | 5aix | 0.475862 | -8.76 | c1cccc(c1)c1ccc(C(=O)N[C@@H]2CC[N@@H+](C)CC2)cn1 | 22 |
| 3n4c_ligand_1_2.mol2 | 3n4c | 0.471014 | -6.65 | c12c([n+](c[nH]1)C)cc(nc2)c1ccccc1 | 16 |
| 4ejn_ligand_2_7.mol2 | 4ejn | 0.469388 | -8.55 | c1(ccccc1)[n+]1c2c(ccc(c3ccccc3)n2)[nH]c1 | 21 |
| 3n4c_ligand_2_13.mol2 | 3n4c | 0.467532 | -6.72 | c12c([n+](c[nH]1)C)cc(nc2C=N)c1ccccc1 | 18 |
| 4i8x_ligand.mol2 | 4i8x | 0.464 | -7.38 | c1(ccccc1)c1ncc(cc1)C(=O)O | 16 |
101 ,
11

