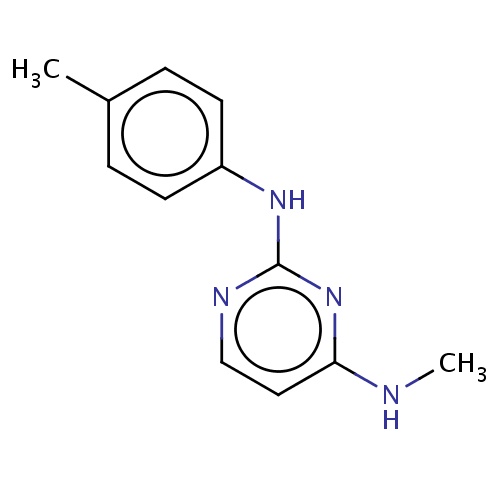
Common name
N4-methyl-N2-(p-tolyl)pyrimidine-2,4-diamine
IUPAC name
N4-methyl-N2-(p-tolyl)pyrimidine-2,4-diamine
SMILES
n1c(ccnc1Nc2ccc(cc2)C)NC
Common name
N4-methyl-N2-(p-tolyl)pyrimidine-2,4-diamine
IUPAC name
N4-methyl-N2-(p-tolyl)pyrimidine-2,4-diamine
SMILES
n1c(ccnc1Nc2ccc(cc2)C)NC
INCHI
InChI=1S/C12H14N4/c1-9-3-5-10(6-4-9)15-12-14-8-7-11(13-2)16-12/h3-8H,1-2H3,(H2,13,14,15,16)
FORMULA
C12H14N4

Common name
N4-methyl-N2-(p-tolyl)pyrimidine-2,4-diamine
IUPAC name
N4-methyl-N2-(p-tolyl)pyrimidine-2,4-diamine
Molecular weight
214.266
clogP
1.812
clogS
-4.146
Frequency
0.0003
HBond Acceptor
2
HBond Donor
2
Total PolarSurface Area
49.84
Number of Rings
2
Rotatable Bond
3
| Drug ID | Common name | Structure CAS | Compound class | Therapeutic area |
|---|---|---|---|---|
| FDBD01419 | Pazopanib |

|
Antineoplastic Agents; Immunosuppressive Agents; Protein Kinase Inhibitors; Antineoplastic and Immunomodulating Agents; Cytochrome P-450 CYP1A2 Inhibitors; Cytochrome P-450 CYP1A2 Inducers; Cytochrome P-450 CYP2C8 Inhibitors; Cytochrome P-450 CYP2C8 Inducers; CYP2D6 Inducers; CYP2D6 Inducers (strong); CYP3A4 Inhibitors; | Treatment of advanced renal cell cancer and advanced soft tissue sarcoma (in patients previously treated with chemotherapy) . |
1 ,
1
| FRAGNAME | PDBID | SIMILIRITY | XSCORE | SMILE | HAC |
|---|---|---|---|---|---|
| 4im0_ligand_5_447.mol2 | 4im0 | 0.909091 | -7.67 | Cc1cc(Nc2nc(ccn2)NC)ccc1 | 16 |
| 4im0_ligand_4_299.mol2 | 4im0 | 0.887097 | -7.55 | c1(nc(ncc1)Nc1ccccc1)NC | 15 |
| 4dea_ligand.mol2 | 4dea | 0.805195 | -9.18 | C(=O)(O)c1ccc(Nc2nc(Nc3ccc(cc3)C(=O)O)ncc2)cc1 | 27 |
| 1z5m_ligand_4_1.mol2 | 1z5m | 0.753425 | -7.93 | c1(ccccc1)Nc1nc(c(cn1)Br)NC | 16 |
| 3exo_ligand_2_1.mol2 | 3exo | 0.75 | -7.33 | c1(ccncn1)Nc1ccc(cc1)C | 14 |
116 ,
12

