
Common name
1-pyrrolidin-1-ylethanone
IUPAC name
1-pyrrolidin-1-ylethanone
SMILES
CC(=O)N1CCCC1
Common name
1-pyrrolidin-1-ylethanone
IUPAC name
1-pyrrolidin-1-ylethanone
SMILES
CC(=O)N1CCCC1
INCHI
InChI=1S/C6H11NO/c1-6(8)7-4-2-3-5-7/h2-5H2,1H3
FORMULA
C6H11NO

Common name
1-pyrrolidin-1-ylethanone
IUPAC name
1-pyrrolidin-1-ylethanone
Molecular weight
113.158
clogP
0.947
clogS
-0.603
Frequency
0.0010
HBond Acceptor
1
HBond Donor
0
Total PolarSurface Area
20.31
Number of Rings
1
Rotatable Bond
1
| Drug ID | Common name | Structure CAS | Compound class | Therapeutic area |
|---|---|---|---|---|
| FDBD01682 | Daclatasvir |

|
Antiinfectives for Systemic Use; Direct Acting Antivirals; Antivirals for Systemic Use; CYP3A4 Inhibitors; | Daklinza is used to treat patients who have chronic hepatitis C virus (HCV) genotype 3 infection. Daklinza is typically taken in conjunction with sofosbuvir. (2). |
| FDBD01793 | Ombitasvir |
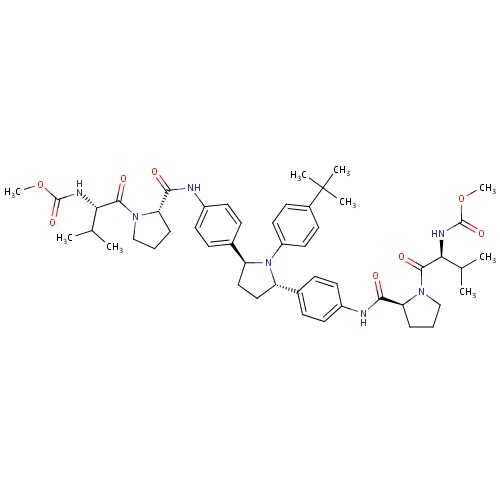
|
Antiinfectives for Systemic Use; Direct Acting Antivirals; Antivirals for Systemic Use; | For use in combination with paritaprevir, ritonavir and dasabuvir for the treatment of HCV genotype 1, and with paritaprevir and ritonavir for the treatment of HCV genotype 4. |
| FDBD01836 | Elbasvir |
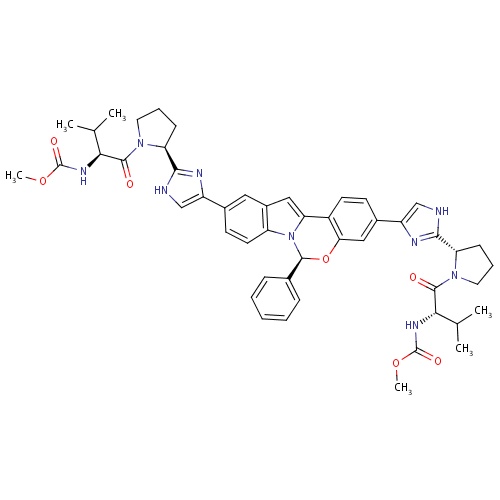
|
; |
3 ,
1
| FRAGNAME | PDBID | SIMILIRITY | XSCORE | SMILE | HAC |
|---|---|---|---|---|---|
| 4jlg_ligand_1_3.mol2 | 4jlg | 1 | -6.52 | C(=O)(N1CCCC1)C | 8 |
| 2i03_ligand_1_4.mol2 | 2i03 | 1 | -6.50 | C1N(CCC1)C(=O)C | 8 |
| 3hpt_ligand_1_0.mol2 | 3hpt | 1 | -6.28 | CC(=O)N1CCCC1 | 8 |
| 3ens_ligand_1_3.mol2 | 3ens | 1 | -6.27 | CC(=O)N1CCCC1 | 8 |
| 4r8y_ligand_2_11.mol2 | 4r8y | 1 | -6.26 | CC(=O)N1C[C@@H](C)CC1 | 9 |
| 1doj_ligand_1_1.mol2 | 1doj | 1 | -6.23 | CC(=O)N1CCCC1 | 8 |
| 3aho_ligand_1_8.mol2 | 3aho | 1 | -6.19 | C(=O)(N1CCCC1)C | 8 |
147 ,
15

