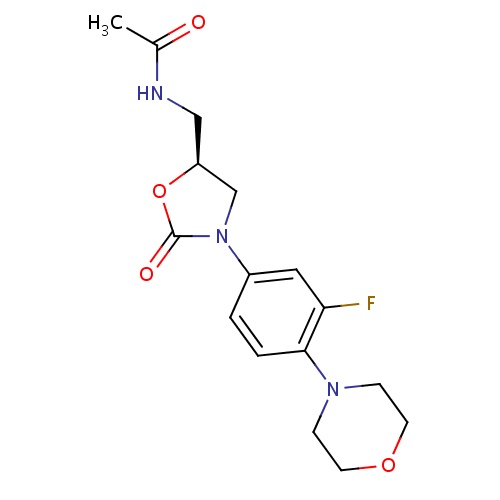
IUPAC name
N-{[(5S)-3-[3-fluoro-4-(morpholin-4-yl)phenyl]-2-oxo-1,3-oxazolidin-5-yl]methyl}acetamide
SMILES
CC(=O)NC[C@H]1CN(C(=O)O1)C1=CC(F)=C(C=C1)N1CCOCC1
Compound class
Anti-Bacterial Agents; Protein Synthesis Inhibitors; Antibacterials for Systemic Use; Antiinfectives for Systemic Use;
Therapeutic area
For the treatment of bacterial infections caused by susceptible strains of vancomycin resistant .
Common name
Linezolid
IUPAC name
N-{[(5S)-3-[3-fluoro-4-(morpholin-4-yl)phenyl]-2-oxo-1,3-oxazolidin-5-yl]methyl}acetamide
SMILES
CC(=O)NC[C@H]1CN(C(=O)O1)C1=CC(F)=C(C=C1)N1CCOCC1
INCHI
InChI=1S/C16H20FN3O4/c1-11(21)18-9-13-10-20(16(22)24-13)12-2-3-15(14(17)8-12)19-4-6-23-7-5-19/h2-3,8,13H,4-7,9-10H2,1H3,(H,18,21)/t13-/m0/s1
FORMULA
C16H20FN3O4

Common name
Linezolid
IUPAC name
N-{[(5S)-3-[3-fluoro-4-(morpholin-4-yl)phenyl]-2-oxo-1,3-oxazolidin-5-yl]methyl}acetamide
Molecular weight
337.346
clogP
1.253
clogS
-2.735
HBond Acceptor
5
HBond Donor
1
Total Polar Surface Area
71.11
Number of Rings
3
Rotatable Bond
4
| Drug ID | Common name | Structure CAS | SMILE | Frequency |
|---|---|---|---|---|
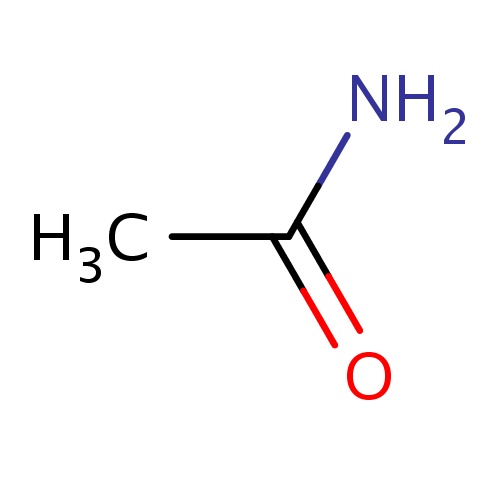
| FDBF00015 | acetamide |
|
NC(=O)C | 0.0460 |
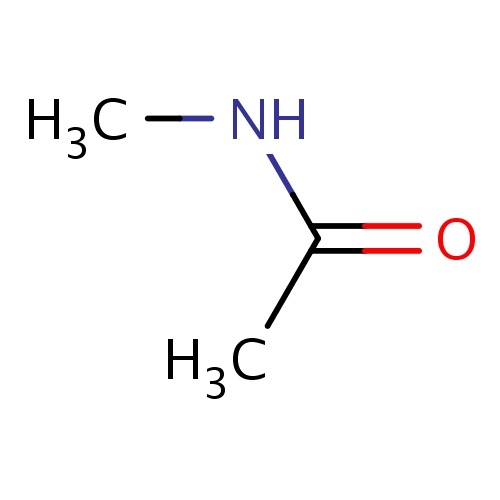
| FDBF00027 | N-methylacetamide |
|
N(C(=O)C)C | 0.0175 |
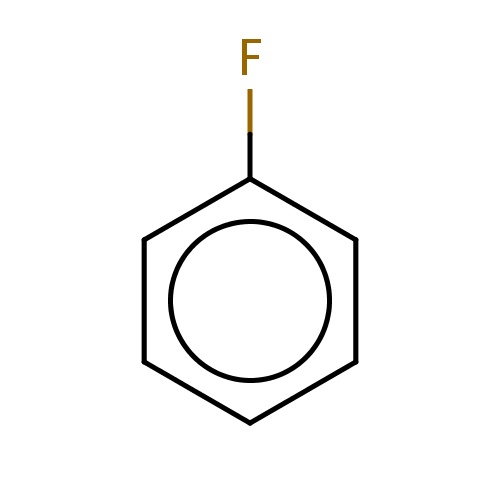
| FDBF00378 | fluorobenzene |
|
Fc1ccccc1 | 0.0237 |
| FDBF00484 | morpholine |

|
O1CCNCC1 | 0.0082 |
| FDBF00707 | oxazolidin-2-one |
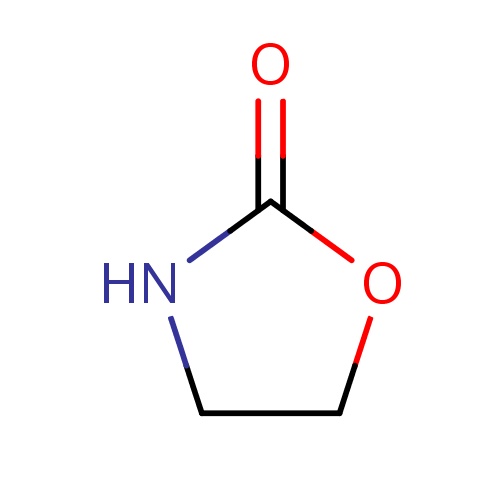
|
O1CCNC1=O | 0.0024 |
| FDBF01339 | 3-(3-fluorophenyl)oxazolidin-2-one |
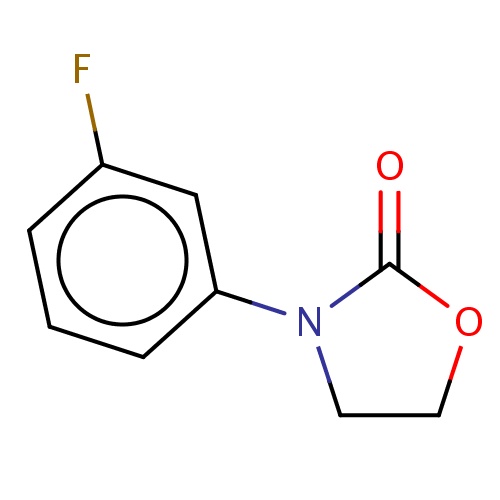
|
O1CCN(C1=O)c2cccc(c2)F | 0.0007 |
| FDBF01340 | 4-(2-fluorophenyl)morpholine |
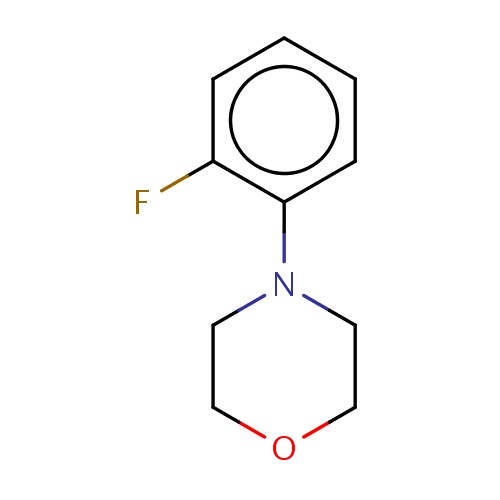
|
O1CCN(CC1)c2c(cccc2)F | 0.0003 |
| FDBF01341 | (5S)-5-methyloxazolidin-2-one |
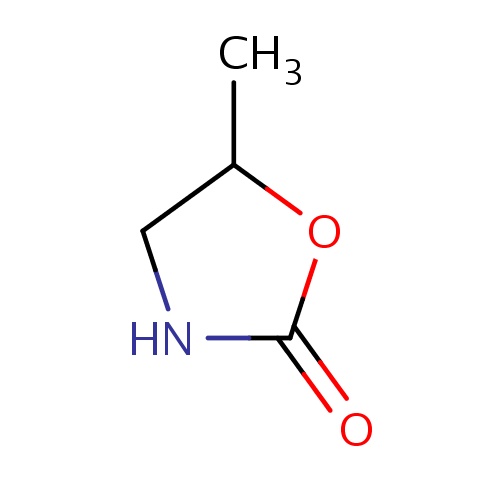
|
CC1OC(=O)NC1 | 0.0014 |
| FDBF01342 | (5S)-3-(3-fluorophenyl)-5-methyl-oxazolidin-2-one |
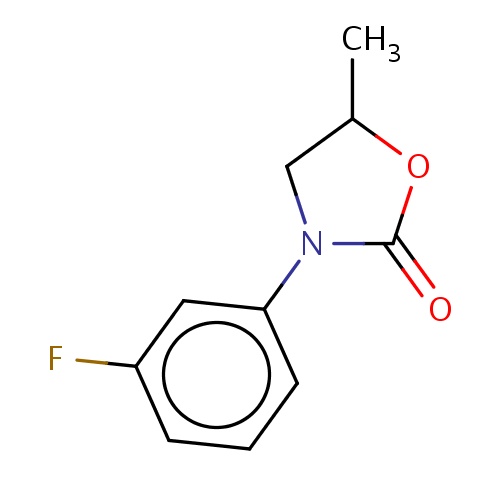
|
CC1OC(=O)N(C1)c2cccc(c2)F | 0.0007 |
| FDBF01343 | N-[[(5S)-3-(3-fluorophenyl)-2-oxo-oxazolidin-5-yl]methyl]acetamide |
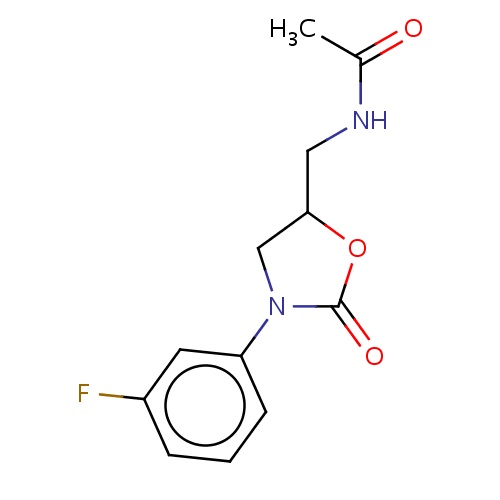
|
C(NC(=O)C)C1OC(=O)N(C1)c2cccc(c2)F | 0.0003 |

