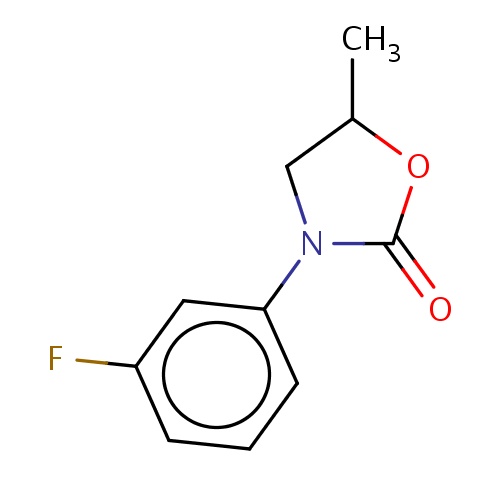
Common name
(5S)-3-(3-fluorophenyl)-5-methyl-oxazolidin-2-one
IUPAC name
(5S)-3-(3-fluorophenyl)-5-methyl-oxazolidin-2-one
SMILES
CC1OC(=O)N(C1)c2cccc(c2)F
Common name
(5S)-3-(3-fluorophenyl)-5-methyl-oxazolidin-2-one
IUPAC name
(5S)-3-(3-fluorophenyl)-5-methyl-oxazolidin-2-one
SMILES
CC1OC(=O)N(C1)c2cccc(c2)F
INCHI
InChI=1S/C10H10FNO2/c1-7-6-12(10(13)14-7)9-4-2-3-8(11)5-9/h2-5,7H,6H2,1H3/t7-/m0/s1
FORMULA
C10H10FNO2

Common name
(5S)-3-(3-fluorophenyl)-5-methyl-oxazolidin-2-one
IUPAC name
(5S)-3-(3-fluorophenyl)-5-methyl-oxazolidin-2-one
Molecular weight
195.190
clogP
1.846
clogS
-2.027
Frequency
0.0007
HBond Acceptor
2
HBond Donor
0
Total PolarSurface Area
29.54
Number of Rings
2
Rotatable Bond
1
| Drug ID | Common name | Structure CAS | Compound class | Therapeutic area |
|---|---|---|---|---|
| FDBD00466 | Linezolid |
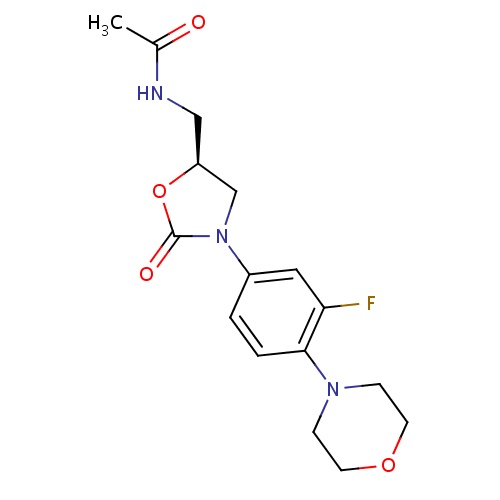
|
Anti-Bacterial Agents; Protein Synthesis Inhibitors; Antibacterials for Systemic Use; Antiinfectives for Systemic Use; | For the treatment of bacterial infections caused by susceptible strains of vancomycin resistant . |
| FDBD01643 | Tedizolid Phosphate |
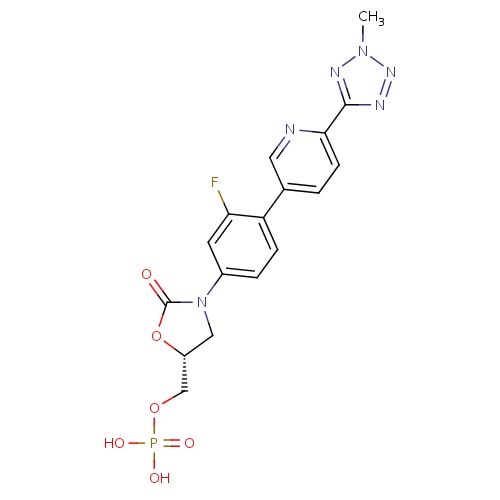
|
Anti-Bacterial Agents; Antibacterials for Systemic Use; Antiinfectives for Systemic Use; | Tedizolid Phosphate is an oxazolidinone antibacterial drug indicated in adults for the treatment of acute bacterial skin and skin structure infections (ABSSSI) caused by susceptible isolates of the following Gram-positive microorganisms: Staphylococcus aureus (including methicillin-resistant [MRSA] and methicillin-susceptible [MSSA] isolates), Streptococcus pyogenes, Streptococcus agalactiae, Streptococcus anginosus Group (including Streptococcus anginosus, Streptococcus intermedius, and Streptococcus constellatus), and Enterococcus faecalis. |
2 ,
1
| FRAGNAME | PDBID | SIMILIRITY | XSCORE | SMILE | HAC |
|---|---|---|---|---|---|
| 4k4f_ligand_1_1.mol2 | 4k4f | 0.853333 | -8.01 | C1C(OC(=O)N1c1ccccc1)(C)C | 14 |
| 4n4t_ligand_1_1.mol2 | 4n4t | 0.853333 | -7.81 | c1c(cccc1)N1CC(OC1=O)(C)C | 14 |
| 2w26_ligand_2_3.mol2 | 2w26 | 0.853333 | -7.01 | c1(ccccc1)N1C(=O)O[C@@H](C)C1 | 13 |
| 2q55_ligand_1_1.mol2 | 2q55 | 0.76 | -6.55 | C1OC(=O)N(C1)c1ccccc1 | 12 |
| 2i0a_ligand_1_1.mol2 | 2i0a | 0.76 | -5.92 | N1(C(=O)OCC1)c1ccccc1 | 12 |
| 3gi5_ligand_1_1.mol2 | 3gi5 | 0.76 | -5.91 | c1cccc(c1)N1C(=O)OCC1 | 12 |
| 3mxe_ligand_1_9.mol2 | 3mxe | 0.76 | -5.90 | N1(CCOC1=O)c1ccccc1 | 12 |
| 3gi4_ligand_1_10.mol2 | 3gi4 | 0.76 | -5.87 | c1(ccccc1)N1C(=O)OCC1 | 12 |
| 3gi6_ligand_1_11.mol2 | 3gi6 | 0.76 | -5.86 | c1(ccccc1)N1C(=O)OCC1 | 12 |
| 2q55_ligand_3_0.mol2 | 2q55 | 0.680851 | -6.84 | [C@@H]1(OC(=O)N(C1)c1ccccc1)C(=O)NC | 16 |
100 ,
11

