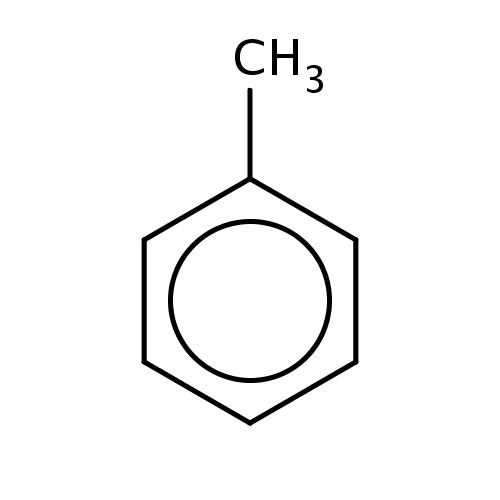
Common name
toluene
IUPAC name
toluene
SMILES
c1(ccccc1)C
Common name
toluene
IUPAC name
toluene
SMILES
c1(ccccc1)C
INCHI
InChI=1S/C7H8/c1-7-5-3-2-4-6-7/h2-6H,1H3
FORMULA
C7H8

Common name
toluene
IUPAC name
toluene
Molecular weight
92.138
clogP
2.443
clogS
-1.933
Frequency
0.1268
HBond Acceptor
0
HBond Donor
0
Total PolarSurface Area
0
Number of Rings
1
Rotatable Bond
0
| Drug ID | Common name | Structure CAS | Compound class | Therapeutic area |
|---|---|---|---|---|
| FDBD00209 | Tolcapone |

|
Antidyskinetics; Nervous System; Anti-Parkinson Drugs; Dopaminergic Agents; Cytochrome P-450 CYP2C9 Inhibitors; Cytochrome P-450 CYP2C9 Inducers; | Used as an adjunct to levodopa/carbidopa therapy for the symptomatic treatment of Parkinson's Disease. This drug is generally reserved for patients with parkinsonian syndrome receiving levodopa/carbidopa who are experiencing symptom fluctuations and are not responding adequately to or are not candidates for other adjunctive therapies. |
| FDBD00218 | Methadone |
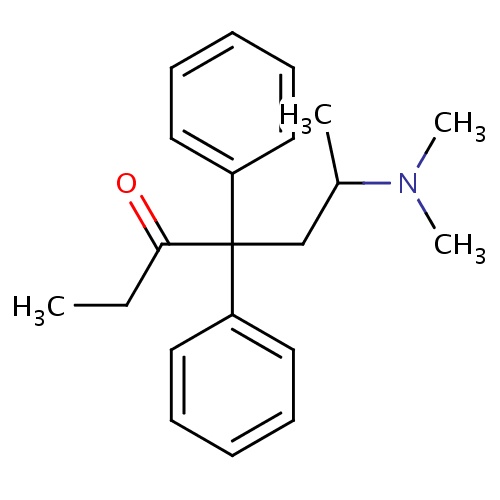
|
Analgesics; Analgesics, Opioid; Narcotics; Antitussive Agents; Nervous System; Drugs Used in Addictive Disorders; Opioids; Diphenylpropylamine Derivatives; Drugs Used in Opioid Dependence; Cytochrome P-450 CYP1A2 Inhibitors; Cytochrome P-450 CYP2C9 Inhibitors; Cytochrome P-450 CYP1A2 Inducers; Cytochrome P-450 CYP2C8 Inhibitors; Cytochrome P-450 CYP2C9 Inducers; Cytochrome P-450 CYP2C19 Inducers; Cytochrome P-450 CYP2C8 Inducers; Cytochrome P-450 CYP2B6 Inducers; Cytochrome P-450 CYP2B6 Inhibitors; CYP2B6 Inhibitors (strong); CYP2D6 Inducers; CYP2D6 Inducers (strong); CYP3A4 Inhibitors; Antiemetics Antagonists; Combined Inhibitors of CYP3A4 and P-glycoprotein; | For the treatment of dry cough, drug withdrawal syndrome, opioid type drug dependence, and pain. |
| FDBD00220 | Atenolol |
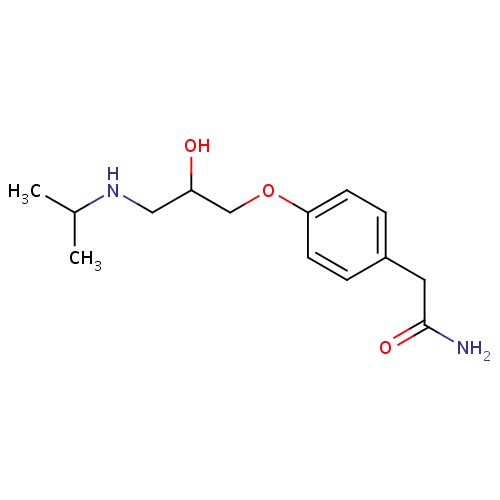
|
Antihypertensive Agents; Adrenergic beta-1 Receptor Antagonists; Sympatholytics; Anti-Arrhythmia Agents; Cardiovascular System; Beta Blocking Agents, Selective; Beta Blocking Agents; Beta Blocking Agents, Selective, and Thiazides; Beta Blocking Agents and Thiazides; | For the management of hypertention and long-term management of patients with angina pectoris. |
| FDBD00226 | Cetirizine |
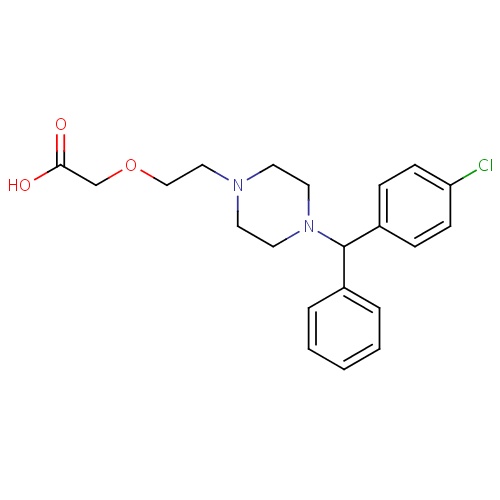
|
Anti-Allergic Agents; Histamine H1 Antagonists, Non-Sedating; Histamine Antagonists; Respiratory System; Antihistamines for Systemic Use; Piperazine Derivatives; | For the relief of symptoms associated with seasonal allergic rhinitis, perennial allergic rhinitis and the treatment of the uncomplicated skin manifestations of chronic idiopathic urticaria. |
| FDBD00238 | Buclizine |
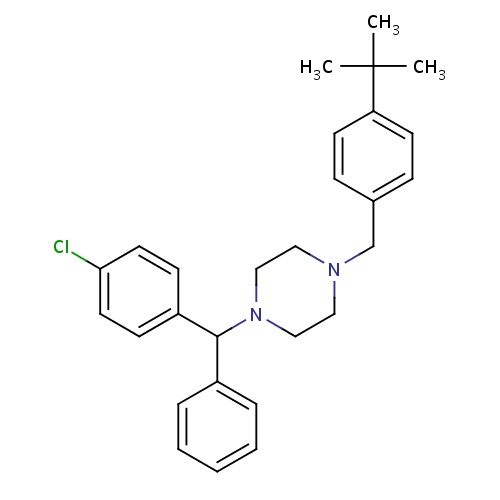
|
Cholinergic Antagonists; Histamine Antagonists; Respiratory System; Antihistamines for Systemic Use; Piperazine Derivatives; | For prevention and treatment of nausea, vomiting, and dizziness associated with motion sickness and vertigo (dizziness caused by other medical problems). |
| FDBD00291 | Rosiglitazone |
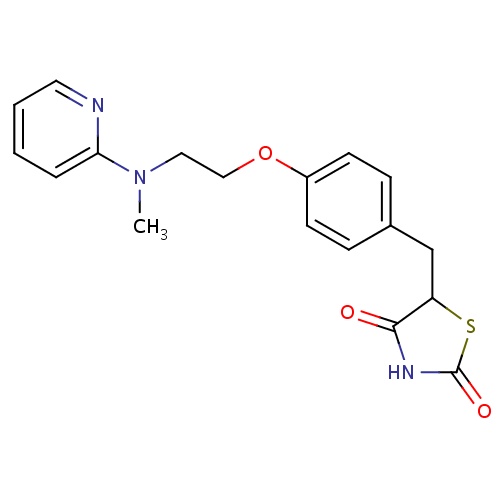
|
Hypoglycemic Agents; Drugs Used in Diabetes; Alimentary Tract and Metabolism; Thiazolidinediones; Blood Glucose Lowering Drugs, Excl. Insulins; Cytochrome P-450 CYP1A2 Inhibitors; Cytochrome P-450 CYP2C9 Inhibitors; Cytochrome P-450 CYP1A2 Inducers; Cytochrome P-450 CYP2C8 Inhibitors; Cytochrome P-450 CYP2C9 Inducers; Cytochrome P-450 CYP2C19 Inducers; Cytochrome P-450 CYP2C8 Inducers; CYP2A6 Inhibitors; CYP2A6 Inhibitors (strong); CYP2A6 Inhibitors (moderate); CYP2A6 Inducers; CYP2A6 Inducers (strong); CYP2D6 Inducers; CYP2D6 Inducers (strong); | Rosiglitazone is indicated as an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes mellitus. |
| FDBD00299 | Methylphenidate |
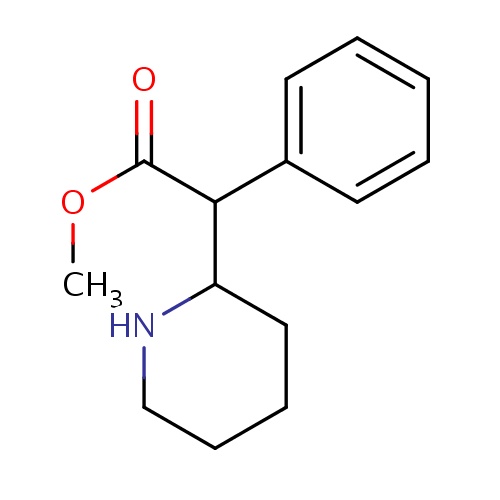
|
Central Nervous System Stimulants; Dopamine Uptake Inhibitors; Nervous System; Psychoanaleptics; Centrally Acting Sympathomimetics; Psychostimulants, Agents Used for Adhd and Nootropics; CYP2D6 Inducers; CYP2D6 Inducers (strong); | For use as an integral part of a total treatment program which typically includes other remedial measures (psychological, educational, social) for a stabilizing effect in children with a behavioral syndrome characterized by the following group of developmentally inappropriate symptoms: moderate-to-severe distractibility, short attention span, hyperactivity, emotional lability, and impulsivity. |
| FDBD00302 | Zolpidem |

|
Hypnotics and Sedatives; GABA-A Receptor Agonists; Nervous System; Psycholeptics; Benzodiazepine Related Drugs; Cytochrome P-450 CYP1A2 Inhibitors; Cytochrome P-450 CYP2C9 Inhibitors; Cytochrome P-450 CYP1A2 Inducers; Cytochrome P-450 CYP2C9 Inducers; Cytochrome P-450 CYP2C19 Inducers; CYP2D6 Inducers; CYP2D6 Inducers (strong); CYP3A4 Inhibitors; | For the short-term treatment of insomnia. |
| FDBD00304 | Triprolidine |
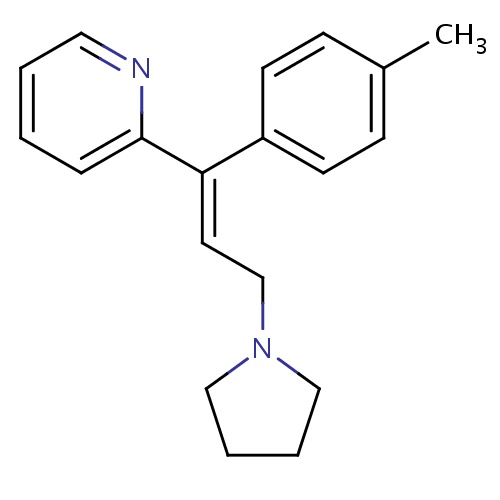
|
Anti-Allergic Agents; Histamine H1 Antagonists; Respiratory System; Antihistamines for Systemic Use; CYP2D6 Inducers; CYP2D6 Inducers (strong); | For the symptomatic relief of seasonal or perennial allergic rhinitis or nonallergic rhinitis; allergic conjunctivitis; and mild, uncomplicated allergic skin manifestations of urticaria and angioedema. Also used in combination with other agents for the symptomatic relief of symptoms associated with the common cold. |
| FDBD00313 | Bendroflumethiazide |
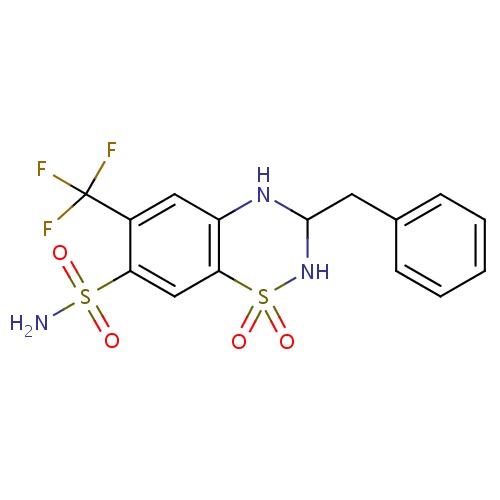
|
Antihypertensive Agents; Diuretics; Sodium Chloride Symporter Inhibitors; Cardiovascular System; Thiazides, Plain; Low-Ceiling Diuretics, Thiazides; Low-Ceiling Diuretics and Potassium-Sparing Agents; | For the treatment of high blood pressure and management of edema related to heart failure. |
369 ,
37
| FRAGNAME | PDBID | SIMILIRITY | XSCORE | SMILE | HAC |
|---|---|---|---|---|---|
| 4i7k_ligand_frag_0.mol2 | 4i7k | 1 | -7.31 | Cc1ccccc1 | 7 |
| 4i7k_ligand.mol2 | 4i7k | 1 | -7.31 | Cc1ccccc1 | 8 |
| 4w53_ligand_frag_0.mol2 | 4w53 | 1 | -7.31 | Cc1ccccc1 | 7 |
| 4w53_ligand.mol2 | 4w53 | 1 | -7.31 | Cc1ccccc1 | 8 |
| 1usi_ligand_1_1.mol2 | 1usi | 1 | -7.29 | c1(ccccc1)C | 7 |
| 4w55_ligand_1_1.mol2 | 4w55 | 1 | -7.29 | Cc1ccccc1 | 7 |
| 3gkz_ligand_1_1.mol2 | 3gkz | 1 | -7.24 | Cc1ccccc1 | 7 |
| 4gqp_ligand_1_1.mol2 | 4gqp | 1 | -7.24 | Cc1ccccc1 | 7 |
| 1mh5_ligand_1_0.mol2 | 1mh5 | 1 | -7.22 | Cc1ccccc1 | 7 |
2592 ,
260

