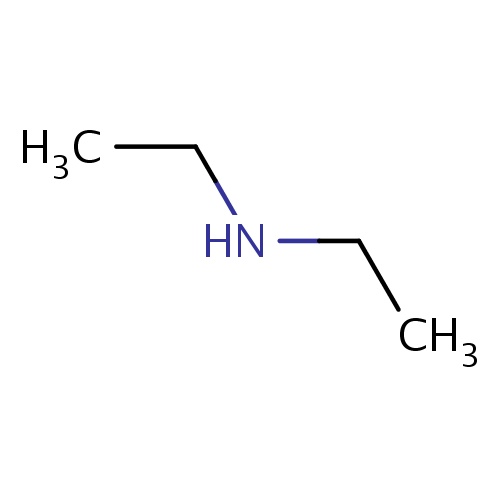
Common name
N-ethylethanamine
IUPAC name
N-ethylethanamine
SMILES
N(CC)CC
Common name
N-ethylethanamine
IUPAC name
N-ethylethanamine
SMILES
N(CC)CC
INCHI
InChI=1S/C4H11N/c1-3-5-4-2/h5H,3-4H2,1-2H3
FORMULA
C4H11N

Common name
N-ethylethanamine
IUPAC name
N-ethylethanamine
Molecular weight
73.137
clogP
0.266
clogS
-1.379
Frequency
0.0240
HBond Acceptor
0
HBond Donor
1
Total PolarSurface Area
12.03
Number of Rings
0
Rotatable Bond
2
| Drug ID | Common name | Structure CAS | Compound class | Therapeutic area |
|---|---|---|---|---|
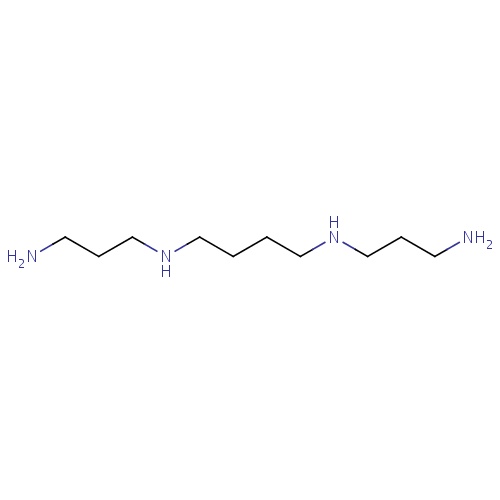
| FDBD00019 | Spermine |
|
Dietary Supplements; Micronutrients; Supplements; | For nutritional supplementation, also for treating dietary shortage or imbalance. |
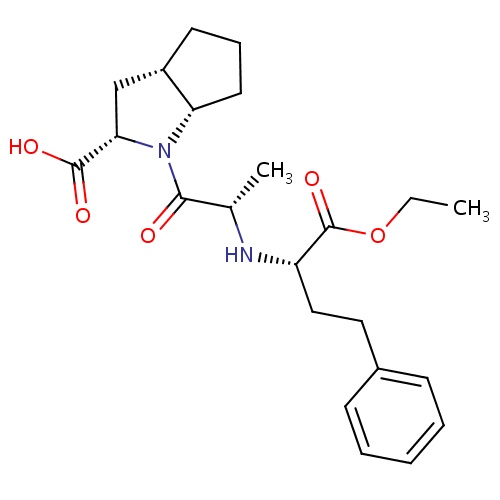
| FDBD00067 | Ramipril |
|
Angiotensin-Converting Enzyme Inhibitors; Antihypertensive Agents; Lipid Modifying Agents; Cardiovascular System; Agents Acting on the Renin-Angiotensin System; ACE Inhibitors, Plain; ACE Inhibitors and Diuretics; ACE Inhibitors and Calcium Channel Blockers; | For the management of mild to severe hypertension. May be used to reduce cardiovascular mortality following myocardial infarction in hemodynamically stable individuals who develop clinical signs of congestive heart failure within a few days following myocardial infarction. To reduce the rate of death, myocardial infarction and stroke in individuals at high risk of cardiovascular events. May be used to slow the progression of renal disease in individuals with hypertension, diabetes mellitus and microalubinuria or overt nephropathy. |
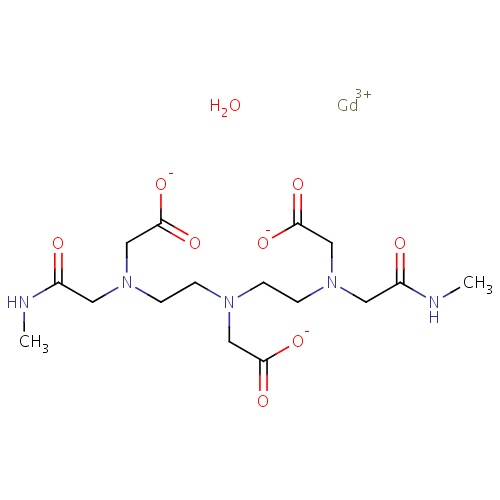
| FDBD00113 | Gadodiamide |
|
Contrast Media; Paramagnetic Contrast Media; Magnetic Resonance Imaging Contrast Media; | For intravenous use in MRI to visualize lesions with abnormal vascularity (or those thought to cause abnormalities in the blood-brain barrier) in the brain (intracranial lesions), spine, and associated tissues. |
| FDBD00168 | Lidocaine |
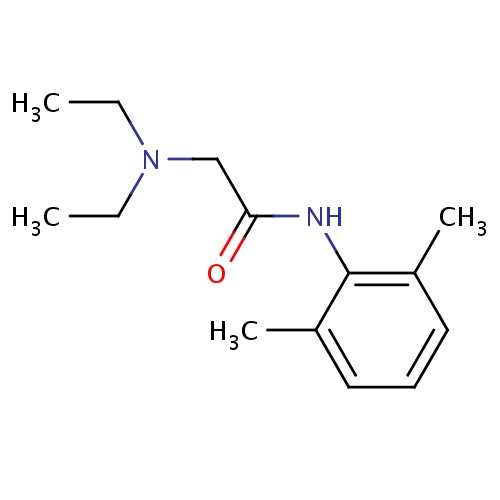
|
Anti-Arrhythmia Agents; Voltage-Gated Sodium Channel Blockers; Anesthetics, Local; Anesthetics; Respiratory System; Ophthalmologicals; Sensory Organs; Throat Preparations; Dermatologicals; Nervous System; Cardiovascular System; Antiarrhythmics, Class I and Iii; Cardiac Therapy; Antiarrythmics, Class I and Iii; Antiarrhythmics, Class Ib; Local Anesthetics; Agents for Treatment of Hemorrhoids and Anal Fissures for Topical Use; Vasoprotectives; Anesthetics for Topical Use; Antipruritics, Incl. Antihistamines, Anesthetics, Etc.; Amides; Analgesics and Anesthetics; Otologicals; Cytochrome P-450 CYP1A2 Inhibitors; Cytochrome P-450 CYP2C9 Inhibitors; Cytochrome P-450 CYP1A2 Inducers; Cytochrome P-450 CYP2C8 Inhibitors; Cytochrome P-450 CYP2C9 Inducers; Cytochrome P-450 CYP2C8 Inducers; Cytochrome P-450 CYP2B6 Inducers; Cytochrome P-450 CYP2B6 Inhibitors; CYP2A6 Inhibitors; CYP2A6 Inhibitors (strong); CYP2A6 Inhibitors (moderate); CYP2A6 Inducers; CYP2A6 Inducers (strong); CYP2B6 Inhibitors (strong); CYP2D6 Inducers; CYP2D6 Inducers (strong); CYP3A4 Inhibitors; Combined Inhibitors of CYP3A4 and P-glycoprotein; | For production of local or regional anesthesia by infiltration techniques such as percutaneous injection and intravenous regional anesthesia by peripheral nerve block techniques such as brachial plexus and intercostal and by central neural techniques such as lumbar and caudal epidural blocks. |
| FDBD00195 | Ibutilide |
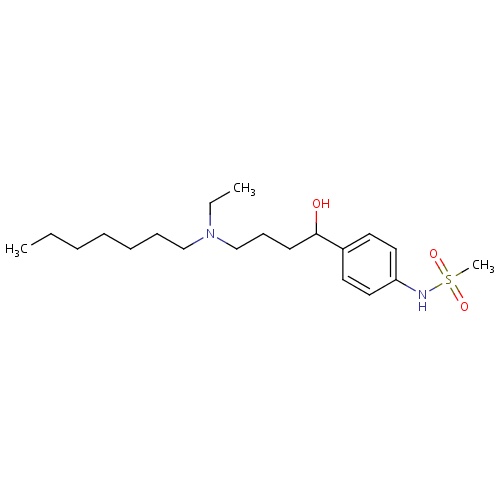
|
Anti-Arrhythmia Agents; Cardiovascular System; Antiarrhythmics, Class III; Antiarrhythmics, Class I and Iii; Cardiac Therapy; Antiarrythmics, Class I and Iii; | Indicated for the rapid conversion of atrial fibrillation or atrial flutter of recent onset to sinus rhythm. |
| FDBD00257 | Colestipol |
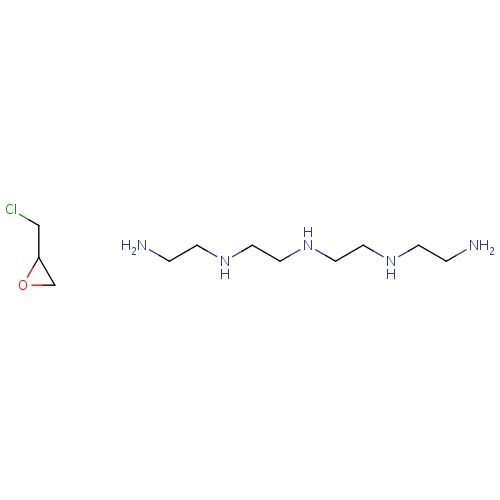
|
Anion Exchange Resins; Hypolipidemic Agents; Bile acid sequestrants; Lipid Modifying Agents, Plain; Lipid Modifying Agents; Cardiovascular System; Sequestering Agents; | For use, as adjunctive therapy to diet, for the reduction of elevated serum total and LDL-C in patients with primary hypercholesterolemia (elevated LDL-C) who do not respond adequately to diet. |
| FDBD00272 | Ethopropazine |
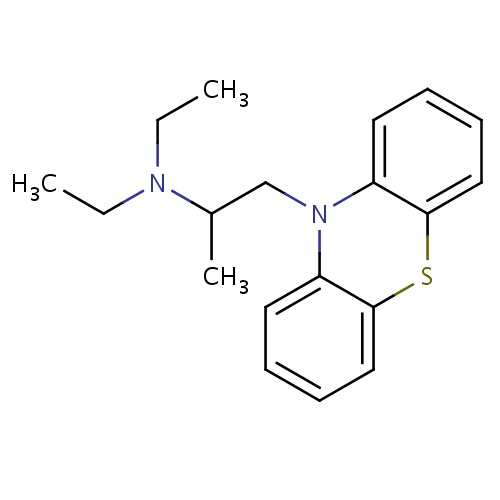
|
Muscarinic Antagonists; Histamine Antagonists; Adrenergic Antagonists; Antidyskinetics; Nervous System; Anti-Parkinson Drugs; Anticholinergics; Tertiary Amines; | For use in the treatment of Parkinson's disease and also used to control severe reactions to certain medicines such as reserpine. |
| FDBD00389 | Trandolapril |
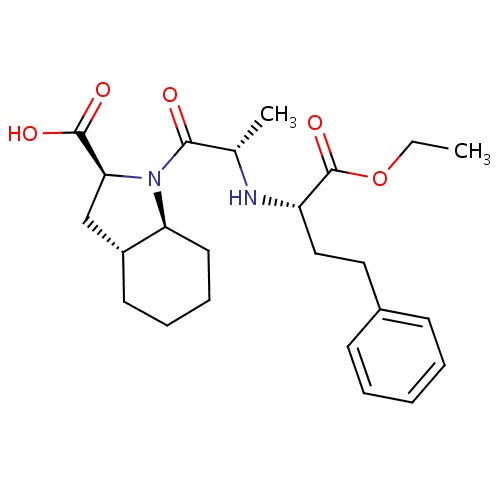
|
Angiotensin-Converting Enzyme Inhibitors; Antihypertensive Agents; Cardiovascular System; Agents Acting on the Renin-Angiotensin System; ACE Inhibitors, Plain; ACE Inhibitors and Calcium Channel Blockers; | For the treatment of mild to moderate hypertension, as an adjunct in the treatment of congestive heart failure (CHF), to improve survival following myocardial infarction (MI) in individuals who are hemodynamically stable and demonstrate symptoms of left ventricular systolic dysfunction or signs of CHF within a few days following acute MI, and to slow progression of renal disease in hypertensive patients with diabetes mellitus and microalbuminuria or overt nephropathy. |
| FDBD00396 | Cinchocaine |
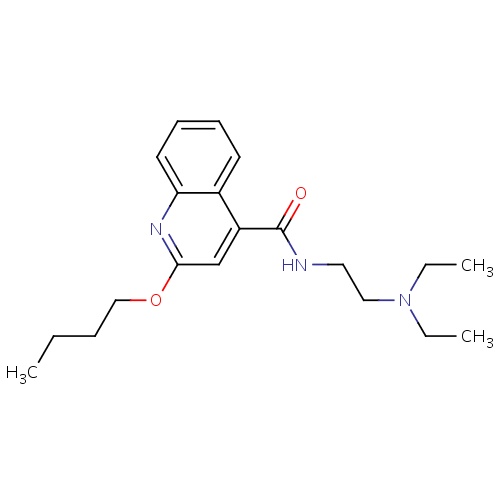
|
Anesthetics, Local; Anesthetics; Ophthalmologicals; Sensory Organs; Dermatologicals; Nervous System; Cardiovascular System; Local Anesthetics; Agents for Treatment of Hemorrhoids and Anal Fissures for Topical Use; Vasoprotectives; Anesthetics for Topical Use; Antipruritics, Incl. Antihistamines, Anesthetics, Etc.; Amides; Analgesics and Anesthetics; Otologicals; | For production of local or regional anesthesia by infiltration techniques such as percutaneous injection and intravenous regional anesthesia by peripheral nerve block techniques such as brachial plexus and intercostal and by central neural techniques such as lumbar and caudal epidural blocks. |
| FDBD00405 | Gadoversetamide |

|
Contrast Media; Paramagnetic Contrast Media; Magnetic Resonance Imaging Contrast Media; | Gadoversetamide is an MRI contrast agent used for MRI diagnostic procedures to provide increased enhancement and visualization of lesions of the brain, spine and liver, including tumors. |
70 ,
8
| FRAGNAME | PDBID | SIMILIRITY | XSCORE | SMILE | HAC |
|---|---|---|---|---|---|
| 1jqe_ligand_4_125.mol2 | 1jqe | 1 | -6.44 | C(C)[NH+](CC)CC | 7 |
| 2q9y_ligand_4_0.mol2 | 2q9y | 1 | -6.43 | C(C)[N+](C)(CC)CC | 8 |
| 4b7z_ligand_4_31.mol2 | 4b7z | 1 | -6.40 | C(C)[NH+](CC)CC | 7 |
| 4b80_ligand_4_31.mol2 | 4b80 | 1 | -6.36 | [NH+](CC)(CC)CC | 7 |
| 4b82_ligand_4_14.mol2 | 4b82 | 1 | -6.36 | C(C)[NH+](CC)CC | 7 |
| 4b81_ligand_4_31.mol2 | 4b81 | 1 | -6.35 | [NH+](CC)(CC)CC | 7 |
| 4b85_ligand_4_14.mol2 | 4b85 | 1 | -6.33 | C(C)[NH+](CC)CC | 7 |
| 2q9y_ligand_3_6.mol2 | 2q9y | 1 | -6.32 | C(C)[NH+](CC)CC | 7 |
| 4b84_ligand_4_30.mol2 | 4b84 | 1 | -6.32 | C(C)[NH+](CC)CC | 7 |
| 4btl_ligand_4_69.mol2 | 4btl | 1 | -6.31 | [NH+](CC)(CC)CC | 7 |
237 ,
24

