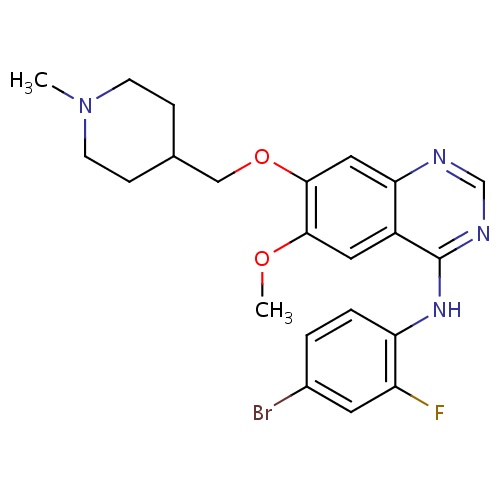
IUPAC name
N-(4-bromo-2-fluorophenyl)-6-methoxy-7-[(1-methylpiperidin-4-yl)methoxy]quinazolin-4-amine
SMILES
COC1=C(OCC2CCN(C)CC2)C=C2N=CN=C(NC3=C(F)C=C(Br)C=C3)C2=C1
Compound class
Antineoplastic Agents; Protein Kinase Inhibitors; Antineoplastic and Immunomodulating Agents; CYP3A4 Inhibitors;
Therapeutic area
Vandetanib is currently approved as an alternative to local therapies for both unresectable and disseminated disease. Because Vandetanib can prolong the Q-T interval, it is contraindicated for use in patients with serious cardiac complications such as congenital long QT syndrome and uncompensated heart failure.
Common name
Vandetanib
IUPAC name
N-(4-bromo-2-fluorophenyl)-6-methoxy-7-[(1-methylpiperidin-4-yl)methoxy]quinazolin-4-amine
SMILES
COC1=C(OCC2CCN(C)CC2)C=C2N=CN=C(NC3=C(F)C=C(Br)C=C3)C2=C1
INCHI
InChI=1S/C22H24BrFN4O2/c1-28-7-5-14(6-8-28)12-30-21-11-19-16(10-20(21)29-2)22(26-13-25-19)27-18-4-3-15(23)9-17(18)24/h3-4,9-11,13-14H,5-8,12H2,1-2H3,(H,25,26,27)
FORMULA
C22H24BrFN4O2

Common name
Vandetanib
IUPAC name
N-(4-bromo-2-fluorophenyl)-6-methoxy-7-[(1-methylpiperidin-4-yl)methoxy]quinazolin-4-amine
Molecular weight
475.354
clogP
4.306
clogS
-7.123
HBond Acceptor
5
HBond Donor
1
Total Polar Surface Area
59.51
Number of Rings
4
Rotatable Bond
6
| Drug ID | Common name | Structure CAS | SMILE | Frequency |
|---|---|---|---|---|
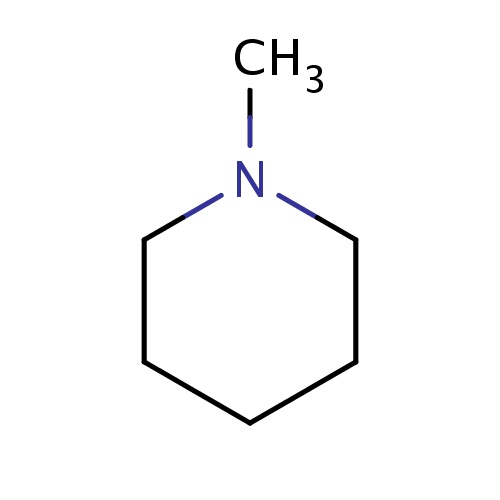
| FDBF00663 | 1-methylpiperidine |
|
CN1CCCCC1 | 0.0172 |
| FDBF00709 | 7-methoxyquinazoline |

|
O(C)c1ccc2c(c1)ncnc2 | 0.0010 |
| FDBF00711 | quinazolin-4-amine |

|
n1c2c(cccc2)c(nc1)N | 0.0031 |
| FDBF00713 | 6-methoxyquinazoline |

|
O(C)c1cc2c(ncnc2)cc1 | 0.0010 |
| FDBF00720 | quinazoline |

|
n1c2c(cccc2)cnc1 | 0.0017 |
| FDBF01199 | quinazolin-7-ol |

|
n1c2c(ccc(c2)O)cnc1 | 0.0010 |
| FDBF01650 | 1,4-dimethylpiperidine |

|
CN1CCC(CC1)C | 0.0021 |
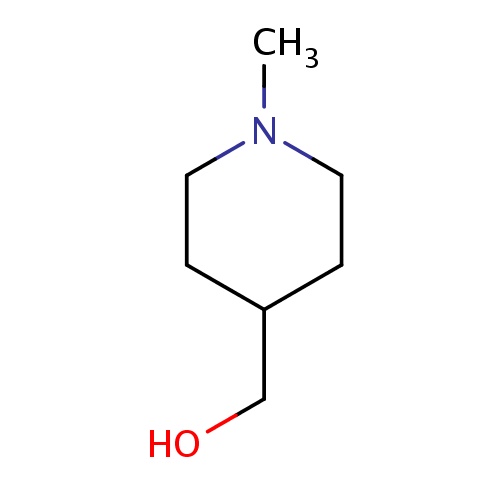
| FDBF02143 | (1-methyl-4-piperidyl)methanol |
|
N1(CCC(CC1)CO)C | 0.0007 |
| FDBF03308 | 4-bromo-2-fluoro-aniline |

|
Nc1c(cc(cc1)Br)F | 0.0003 |
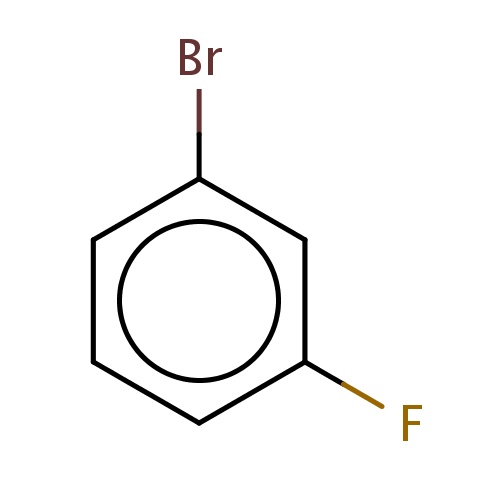
| FDBF03309 | 1-bromo-3-fluoro-benzene |
|
Brc1cc(ccc1)F | 0.0003 |

