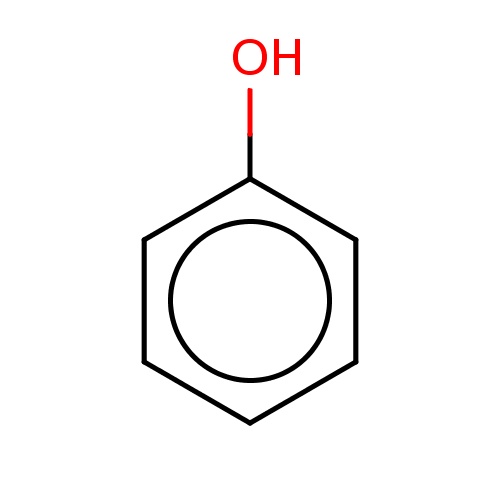
Common name
phenol
IUPAC name
phenol
SMILES
c1ccc(cc1)O
Common name
phenol
IUPAC name
phenol
SMILES
c1ccc(cc1)O
INCHI
InChI=1S/C6H6O/c7-6-4-2-1-3-5-6/h1-5,7H
FORMULA
C6H6O

Common name
phenol
IUPAC name
phenol
Molecular weight
94.111
clogP
1.498
clogS
-0.975
Frequency
0.0897
HBond Acceptor
1
HBond Donor
1
Total PolarSurface Area
20.23
Number of Rings
1
Rotatable Bond
0
| Drug ID | Common name | Structure CAS | Compound class | Therapeutic area |
|---|---|---|---|---|
| FDBD00837 | Formoterol |

|
Sympathomimetics; Adrenergic beta-2 Receptor Agonists; Bronchodilator Agents; Respiratory System; Drugs for Obstructive Airway Diseases; Selective Beta-2-Adrenoreceptor Agonists; Adrenergics, Inhalants; Cytochrome P-450 CYP2C9 Inhibitors; Cytochrome P-450 CYP2C9 Inducers; Cytochrome P-450 CYP2C19 Inducers; CYP2A6 Inhibitors; CYP2A6 Inhibitors (strong); CYP2A6 Inhibitors (moderate); CYP2A6 Inducers; CYP2A6 Inducers (strong); CYP2D6 Inducers; CYP2D6 Inducers (strong); Beta2 Agonists; | For use as long-term maintenance treatment of asthma in patients 6 years of age and older with reversible obstructive airways disease, including patients with symptoms of nocturnal asthma, who are using optimal corticosteroid treatment and experiencing regular or frequent breakthrough symptoms requiring use of a short-acting bronchodilator. Not indicated for asthma that can be successfully managed with occasional use of an inhaled, short-acting beta2-adrenergic agonist. Also used for the prevention of exercise-induced bronchospasm, as well as long-term treatment of bronchospasm associated with COPD. |
| FDBD00855 | Salbutamol |
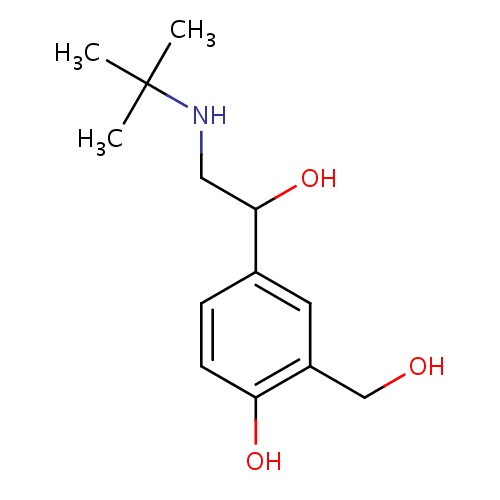
|
Adrenergic beta-2 Receptor Agonists; Bronchodilator Agents; Tocolytic Agents; Respiratory System; Drugs for Obstructive Airway Diseases; Selective Beta-2-Adrenoreceptor Agonists; Adrenergics, Inhalants; Adrenergics for Systemic Use; CYP3A4 Inhibitors; Beta2 Agonists; | For symptomatic relief and prevention of bronchospasm due to bronchial asthma, chronic bronchitis, and other chronic bronchopulmonary disorders such as COPD. |
| FDBD00864 | Edrophonium |

|
Cholinesterase Inhibitors; Antidotes; | For the differential diagnosis of myasthenia gravis and as an adjunct in the evaluation of treatment requirements in this disease. It may also be used for evaluating emergency treatment in myasthenic crises. |
| FDBD00880 | Ketoconazole |

|
Antifungal Agents; 14-alpha Demethylase Inhibitors; Genito Urinary System and Sex Hormones; Antiinfectives for Systemic Use; Dermatologicals; Gynecological Antiinfectives and Antiseptics; Imidazole and Triazole Derivatives; Antifungals for Topical Use; Antifungals for Dermatological Use; Antimycotics for Systemic Use; Imidazole Derivatives; Cytochrome P-450 CYP1A2 Inhibitors; Cytochrome P-450 CYP2C9 Inhibitors; Cytochrome P-450 CYP1A2 Inducers; Cytochrome P-450 CYP2C8 Inhibitors; Cytochrome P-450 CYP3A Inhibitors; Cytochrome P-450 CYP2C9 Inducers; Cytochrome P-450 CYP2C19 Inducers; Cytochrome P-450 CYP2C8 Inducers; Cytochrome P-450 CYP2B6 Inducers; Cytochrome P-450 CYP2B6 Inhibitors; CYP2A6 Inhibitors; CYP2A6 Inhibitors (strong); CYP2A6 Inhibitors (moderate); CYP2A6 Inducers; CYP2A6 Inducers (strong); CYP2B6 Inhibitors (strong); CYP2D6 Inducers; CYP2D6 Inducers (strong); CYP3A4 Inhibitors; BSEP/ABCB11 Inhibitors; Combined Inhibitors of CYP3A4 and P-glycoprotein; | For the treatment of the following systemic fungal infections: candidiasis, chronic mucocutaneous candidiasis, oral thrush, candiduria, blastomycosis, coccidioidomycosis, histoplasmosis, chromomycosis, and paracoccidioidomycosis. |
| FDBD00891 | Fenofibrate |
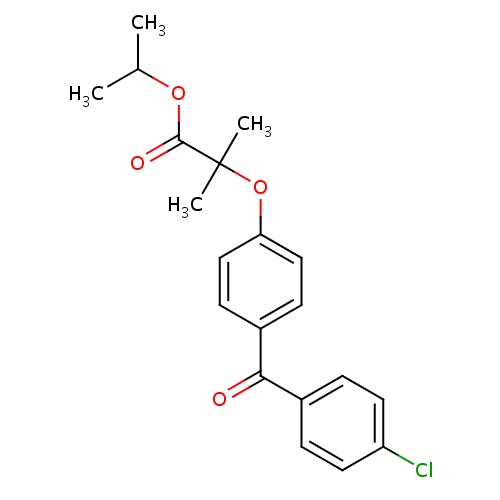
|
Anticholesteremic Agents; Hypolipidemic Agents; Lipid Modifying Agents, Plain; Lipid Modifying Agents; Cardiovascular System; Fibrates; Cytochrome P-450 CYP2C9 Inhibitors; Cytochrome P-450 CYP2C8 Inhibitors; Cytochrome P-450 CYP2C9 Inducers; Cytochrome P-450 CYP2C8 Inducers; CYP3A4 Inhibitors; | For use as adjunctive therapy to diet to reduce elevated LDL-C, Total-C,Triglycerides and Apo B, and to increase HDL-C in adult patients with primary hypercholesterolemia or mixed dyslipidemia (Fredrickson Types IIa and IIb). |
| FDBD00903 | Novobiocin |

|
Enzyme Inhibitors; Anti-Bacterial Agents; Nucleic Acid Synthesis Inhibitors; Antibacterials for Systemic Use; Antiinfectives for Systemic Use; BSEP/ABCB11 Inhibitors; | For the treatment of infections due to staphylococci and other susceptible organisms. |
| FDBD00911 | Amoxicillin |

|
Anti-Bacterial Agents; Penicillins; Genito Urinary System and Sex Hormones; Alimentary Tract and Metabolism; Antibacterials for Systemic Use; Antiinfectives for Systemic Use; Antibacterials for Intramammary Use; Drugs for Peptic Ulcer and Gastro-Oesophageal Reflux Disease (Gord); Drugs for Acid Related Disorders; Beta-Lactam Antibacterials, Penicillins; Beta-Lactam Antibacterials, Penicillins, for Intramammary Use; Penicillins With Extended Spectrum; Antiinfectives and Antiseptics for Intrauterine Use; Antibacterials; Cytochrome P-450 CYP2C19 Inducers; | For the treatment of infections of the ear, nose, and throat, the genitourinary tract, the skin and skin structure, and the lower respiratory tract due to susceptible (only b-lactamase-negative) strains of . |
| FDBD00944 | Hesperetin |

|
Cytochrome P-450 CYP1A2 Inhibitors; Cytochrome P-450 CYP1A2 Inducers; | For lowering cholesterol and, possibly, otherwise favorably affecting lipids. |
| FDBD00952 | Arbutamine |
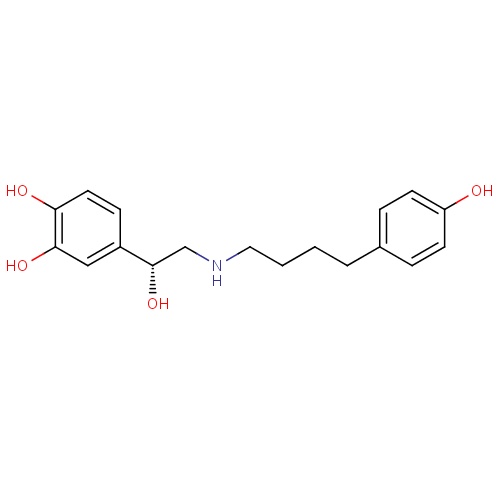
|
Cardiovascular System; Cardiac Therapy; Adrenergic and Dopaminergic Agents; Cardiac Stimulants Excl. Cardiac Glycosides; Beta2 Agonists; | Used to elicit acute cardiovascular responses (cardiac stumulant), similar to those produced by exercise, in order to aid in diagnosing the presence or absence of coronary artery disease (CAD) in patients who cannot exercise adequately. |
| FDBD00981 | Pioglitazone |
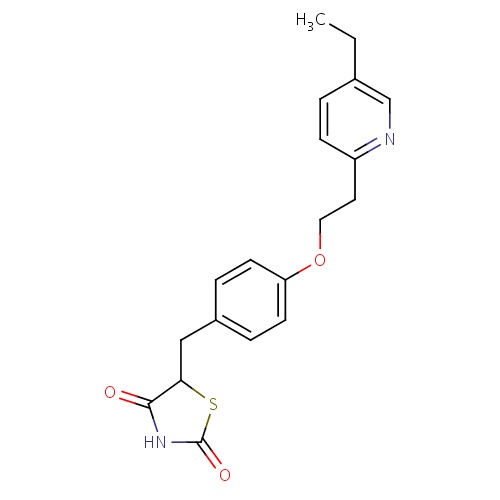
|
Hypoglycemic Agents; Drugs Used in Diabetes; Alimentary Tract and Metabolism; Thiazolidinediones; Blood Glucose Lowering Drugs, Excl. Insulins; Cytochrome P-450 CYP2C9 Inhibitors; Cytochrome P-450 CYP2C8 Inhibitors; Cytochrome P-450 CYP2C9 Inducers; Cytochrome P-450 CYP2C19 Inducers; Cytochrome P-450 CYP2C8 Inducers; CYP2D6 Inducers; CYP2D6 Inducers (strong); CYP3A4 Inhibitors; | Treatment of Type II diabetes mellitus. |
261 ,
27
| FRAGNAME | PDBID | SIMILIRITY | XSCORE | SMILE | HAC |
|---|---|---|---|---|---|
| 4wr8_ligand_frag_0.mol2 | 4wr8 | 1 | -7.04 | c1(ccccc1)O | 7 |
| 4i7l_ligand_frag_0.mol2 | 4i7l | 1 | -7.03 | c1(ccccc1)O | 7 |
| 4i7l_ligand.mol2 | 4i7l | 1 | -7.03 | c1(ccccc1)O | 8 |
| 1mfi_ligand_frag_0.mol2 | 1mfi | 1 | -7.02 | c1ccc(cc1)O | 7 |
| 4wrb_ligand_frag_0.mol2 | 4wrb | 1 | -7.00 | c1(ccccc1)O | 7 |
| 1ljt_ligand_frag_3.mol2 | 1ljt | 1 | -6.98 | c1cc(ccc1)O | 7 |
| 2ra6_ligand_frag_1.mol2 | 2ra6 | 1 | -6.98 | c1ccc(cc1)O | 7 |
| 2ooz_ligand_frag_0.mol2 | 2ooz | 1 | -6.97 | c1(ccccc1)O | 7 |
| 1li2_ligand_frag_0.mol2 | 1li2 | 1 | -6.94 | c1(ccccc1)O | 7 |
1122 ,
113

