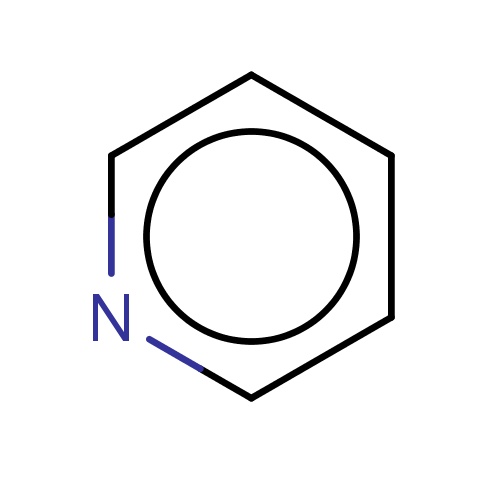
Common name
pyridine
IUPAC name
pyridine
SMILES
c1cccnc1
Common name
pyridine
IUPAC name
pyridine
SMILES
c1cccnc1
INCHI
InChI=1S/C5H5N/c1-2-4-6-5-3-1/h1-5H
FORMULA
C5H5N

Common name
pyridine
IUPAC name
pyridine
Molecular weight
79.100
clogP
1.626
clogS
-1.146
Frequency
0.0333
HBond Acceptor
1
HBond Donor
0
Total PolarSurface Area
12.89
Number of Rings
1
Rotatable Bond
0
| Drug ID | Common name | Structure CAS | Compound class | Therapeutic area |
|---|---|---|---|---|
| FDBD01518 | Chloropyramine |

|
Histamine H1 Antagonists; Histamine Antagonists; Respiratory System; Dermatologicals; Antipruritics, Incl. Antihistamines, Anesthetics, Etc.; Antihistamines for Topical Use; Antihistamines for Systemic Use; Substituted Ethylene Diamines; | For the treatment of allergic conjunctivitis, allergic rhinitis, bronchial asthma, and other atopic (allergic) conditions. |
| FDBD01519 | Dimetindene |
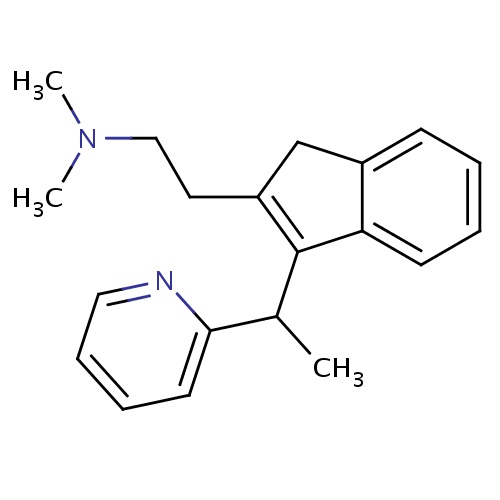
|
Anti-Allergic Agents; Antipruritics; Histamine H1 Antagonists; Cholinergic Antagonists; Histamine Antagonists; Respiratory System; Dermatologicals; Antipruritics, Incl. Antihistamines, Anesthetics, Etc.; Antihistamines for Topical Use; Antihistamines for Systemic Use; Substituted Alkylamines; | Indicated as symptomatic treatment of allergic reactions: urticaria, allergies of the upper respiratory tract such as hey fever and perennial rhinitis, food and drug allergies; pruritus of various origins, except pruritus due to cholestasis; insect bites. Dimethindene is also indicated for pruritus in eruptive skin diseases such as chicken-pox. Dimethindene can also be used as an adjuvant in eczema and other pruriginous dermatoses of allergic origin. |
| FDBD01538 | Vismodegib |

|
Antineoplastic Agents; Antineoplastic and Immunomodulating Agents; Cytochrome P-450 CYP2C9 Inhibitors; Cytochrome P-450 CYP2C8 Inhibitors; Cytochrome P-450 CYP2C9 Inducers; Cytochrome P-450 CYP2C19 Inducers; Cytochrome P-450 CYP2C8 Inducers; CYP3A4 Inhibitors; | Vismodegib is used for treating locally advanced or metastatic basal cell carcinoma in patients whose carcinoma has recurred after surgery, and in patients who are not candidates for surgery or radiation. |
| FDBD01561 | Regorafenib |
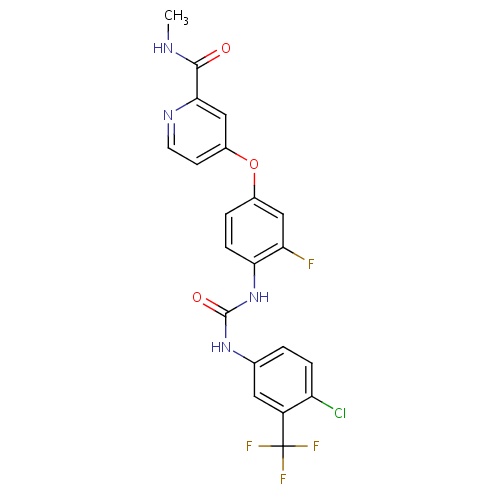
|
Antineoplastic Agents; Protein Kinase Inhibitors; Antineoplastic and Immunomodulating Agents; Cytochrome P-450 CYP2C9 Inhibitors; Cytochrome P-450 CYP2C8 Inhibitors; Cytochrome P-450 CYP2C9 Inducers; Cytochrome P-450 CYP2C19 Inducers; Cytochrome P-450 CYP2C8 Inducers; Cytochrome P-450 CYP2B6 Inducers; Cytochrome P-450 CYP2B6 Inhibitors; CYP2B6 Inhibitors (strong); CYP3A4 Inhibitors; Combined Inhibitors of CYP3A4 and P-glycoprotein; | Regorafenib is indicated for the treatment of patients with metastatic colorectal cancer (CRC) who have been previously treated with fluoropyrimidine-, oxaliplatin- and irinotecan-based chemotherapy, an anti-VEGF therapy, and, if KRAS wild type, an anti-EGFR therapy. Regorafenib is also indicated for the treatment of patients with locally advanced, unresectable or metastatic gastrointestinal stromal tumor (GIST) who have been previously treated with imatinib mesylate and sunitinib malate. |
| FDBD01607 | Etofibrate |
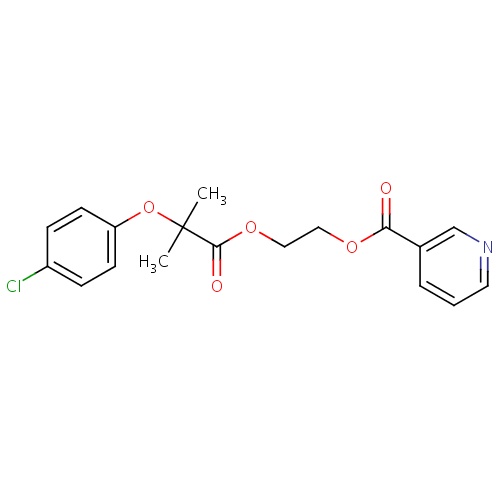
|
Lipid Modifying Agents, Plain; Lipid Modifying Agents; Cardiovascular System; Fibrates; | |
| FDBD01622 | Cephaloridine |
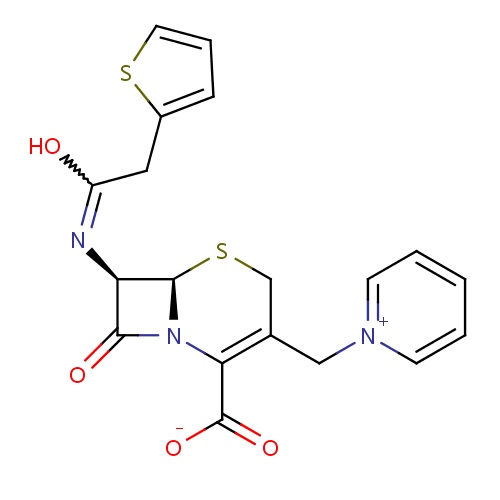
|
Anti-Bacterial Agents; | |
| FDBD01631 | Bisacodyl |

|
Cathartics; Laxatives; Alimentary Tract and Metabolism; Drugs for Constipation; Enemas; Contact Laxatives; | Indicated for cleansing of the colon as a preparation for colonoscopy in adults. |
| FDBD01636 | Vorapaxar |
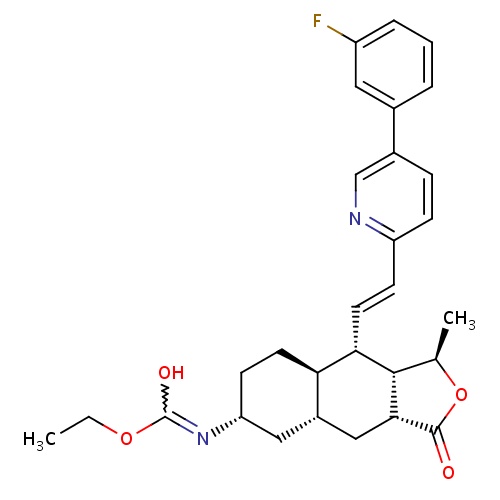
|
Platelet Aggregation Inhibitors; Antithrombotic Agents; Blood and Blood Forming Organs; Platelet Aggregation Inhibitors Excl. Heparin; | Vorapaxar is indicated for the reduction of thrombotic cardiovascular events in patients with a history of myocardial infarction (MI) or peripheral arterial disease (PAD). It is usually co-administered with acetylsalicylic acid (ASA) and/or clopidogrel, and should therefore be administered as an addition to these medications as it has not been studied alone. |
| FDBD01643 | Tedizolid Phosphate |
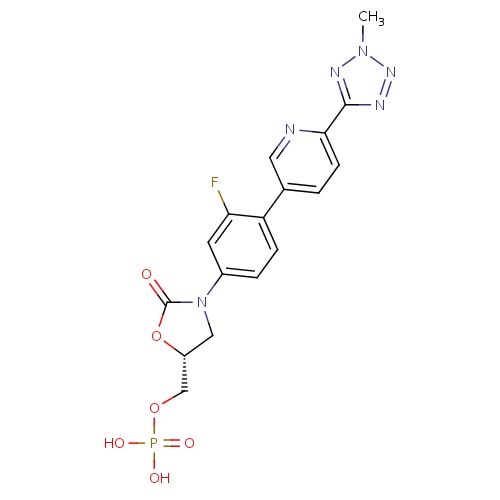
|
Anti-Bacterial Agents; Antibacterials for Systemic Use; Antiinfectives for Systemic Use; | Tedizolid Phosphate is an oxazolidinone antibacterial drug indicated in adults for the treatment of acute bacterial skin and skin structure infections (ABSSSI) caused by susceptible isolates of the following Gram-positive microorganisms: Staphylococcus aureus (including methicillin-resistant [MRSA] and methicillin-susceptible [MSSA] isolates), Streptococcus pyogenes, Streptococcus agalactiae, Streptococcus anginosus Group (including Streptococcus anginosus, Streptococcus intermedius, and Streptococcus constellatus), and Enterococcus faecalis. |
| FDBD01645 | Netupitant |
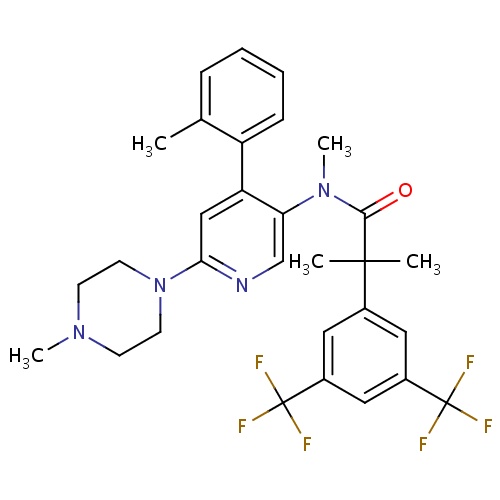
|
Cytochrome P-450 CYP2C9 Inhibitors; Cytochrome P-450 CYP2C9 Inducers; CYP2D6 Inducers; CYP2D6 Inducers (strong); | Netupitant is an antiemitic drug approved by the FDA in October 2014 for use in combination with palonosetron for the prevention of acute and delayed vomiting and nausea associated with cancer chemotherapy including highly emetogenic chemotherapy. |
97 ,
10
| FRAGNAME | PDBID | SIMILIRITY | XSCORE | SMILE | HAC |
|---|---|---|---|---|---|
| 1xh5_ligand_frag_1.mol2 | 1xh5 | 1 | -6.40 | c1ccncc1 | 6 |
| 1xh6_ligand_frag_7.mol2 | 1xh6 | 1 | -6.40 | c1ccncc1 | 6 |
| 1svh_ligand_frag_7.mol2 | 1svh | 1 | -6.39 | c1ccncc1 | 6 |
| 1xh9_ligand_frag_1.mol2 | 1xh9 | 1 | -6.39 | c1ccncc1 | 6 |
| 2gnj_ligand_frag_3.mol2 | 2gnj | 1 | -6.39 | c1ccncc1 | 6 |
| 3dne_ligand_frag_1.mol2 | 3dne | 1 | -6.39 | c1ccncc1 | 6 |
| 1xh4_ligand_frag_1.mol2 | 1xh4 | 1 | -6.38 | c1ccncc1 | 6 |
| 1xh8_ligand_frag_1.mol2 | 1xh8 | 1 | -6.38 | c1ccncc1 | 6 |
645 ,
65

