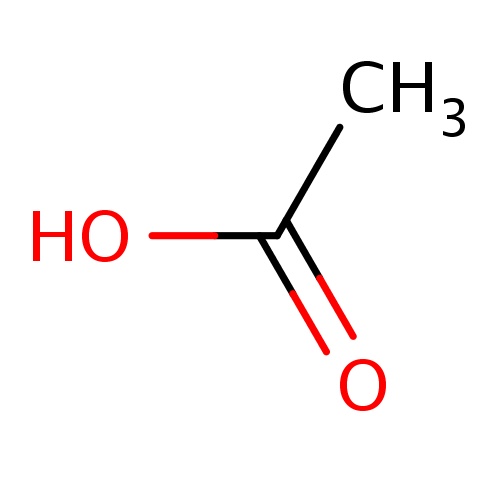
Common name
acetic acid
IUPAC name
acetic acid
SMILES
CC(=O)O
Common name
acetic acid
IUPAC name
acetic acid
SMILES
CC(=O)O
INCHI
InChI=1S/C2H4O2/c1-2(3)4/h1H3,(H,3,4)
FORMULA
C2H4O2

Common name
acetic acid
IUPAC name
acetic acid
Molecular weight
60.052
clogP
-0.484
clogS
0.534
Frequency
0.0687
HBond Acceptor
2
HBond Donor
1
Total PolarSurface Area
37.3
Number of Rings
0
Rotatable Bond
0
| Drug ID | Common name | Structure CAS | Compound class | Therapeutic area |
|---|---|---|---|---|
| FDBD01237 | Ursodeoxycholic acid |

|
Cholagogues and Choleretics; Alimentary Tract and Metabolism; Bile and Liver Therapy; Bile Acid Preparations; Bile Therapy; CYP2E1 Inhibitors; CYP2E1 Inducers; CYP2E1 Inducers (strong); BSEP/ABCB11 Inhibitors; | The drug reduces cholesterol absorption and is used to dissolve (cholesterol) gallstones in patients who want an alternative to surgery. |
| FDBD01257 | Ticarcillin |

|
Anti-Bacterial Agents; Penicillins; Antibacterials for Systemic Use; Antiinfectives for Systemic Use; Beta-Lactam Antibacterials, Penicillins; Penicillins With Extended Spectrum; | For the treatment of bacterial infections. |
| FDBD01289 | Cholic Acid |
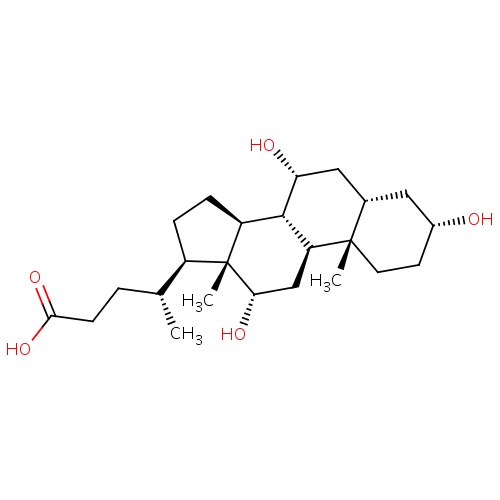
|
Alimentary Tract and Metabolism; Bile and Liver Therapy; Bile Acid Preparations; Bile Therapy; BSEP/ABCB11 Inhibitors; | Oral cholic acid is indicated for: treatment of bile acid synthesis disorders due to single enzyme defects; and as adjunctive treatment of peroxisomal disorders including Zellweger spectrum disorders in patients who exhibit manifestations of liver disease, steatorrhea or complications from decreased fat soluble vitamin absorption. |
| FDBD01295 | Acetic acid |

|
Indicators and Reagents; Anti-Bacterial Agents; Sensory Organs; Genito Urinary System and Sex Hormones; Organic Acids; Gynecological Antiinfectives and Antiseptics; Antiinfectives; Otologicals; | Used to treat infections in the ear canal. |
| FDBD01300 | Deoxycholic Acid |

|
Cholagogues and Choleretics; | For improvement in appearance of moderate to severe fullness associated with submental fat in adults. |
| FDBD01305 | Oleic Acid |
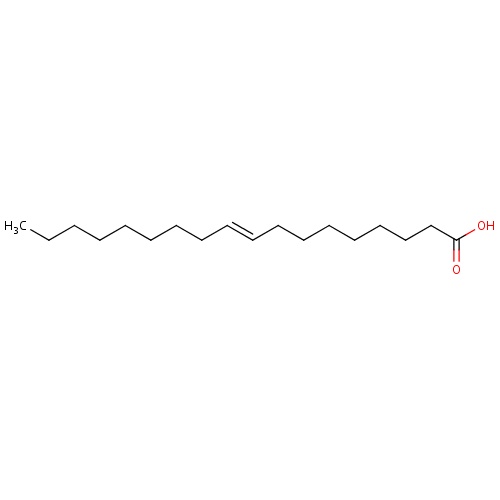
|
; | |
| FDBD01307 | Carbocisteine |

|
Anti-Infective Agents, Local; Expectorants; Mucolytics; Cough and Cold Preparations; Respiratory System; | Used to help relieve the symptoms of chronic obstructive pulmonary disorder (COPD) and bronchiectasis. |
| FDBD01311 | Latamoxef |

|
Anti-Bacterial Agents; Cephalosporins; Antibacterials for Systemic Use; Antiinfectives for Systemic Use; Third-Generation Cephalosporins; | Latamoxef is an oxacephem antibiotic usually grouped with the cephalosporins. It is used to treat bacterial infections. Latamoxef is primarily indicated in conditions like Bone and joint infection, GI infections, Gynecological infections, Meningitis, Respiratory tract infections, Septicaemia, Skin infections, Soft tissue infections, UTI. |
| FDBD01342 | Bepotastine |
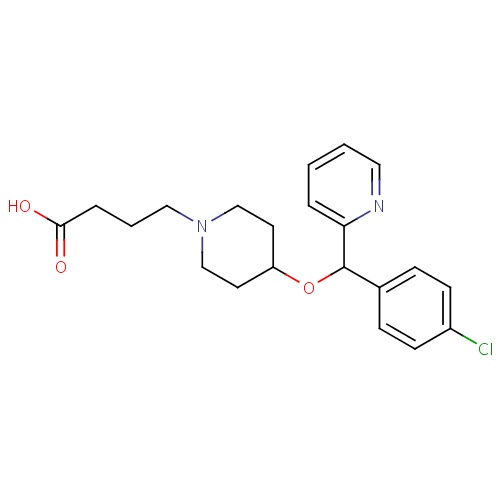
|
Mast Cell Stabilizers; | For the symptomatic treatment of itchy eyes (caused by IgE-induced mast cell degranulation) due to allergic conjunctivitis. |
| FDBD01359 | Trabectedin |

|
Antineoplastic Agents; Immunosuppressive Agents; Antineoplastic Agents, Alkylating; Antineoplastic and Immunomodulating Agents; Cytochrome P-450 CYP2C9 Inhibitors; Cytochrome P-450 CYP2C9 Inducers; Cytochrome P-450 CYP2C19 Inducers; CYP2D6 Inducers; CYP2D6 Inducers (strong); CYP2E1 Inhibitors; CYP2E1 Inducers; CYP2E1 Inducers (strong); CYP3A4 Inhibitors; | Indicated for treatment of advanced soft tissue sarcoma in patients refractory to or unsuitable to receive anthracycline or ifosfamide chemotherapy in Europe, Russia and South Korea. Approved for orphan drug status by the U.S. FDA for treatment of soft tissue sarcomas and ovarian cancer. Investigated for use/treatment in cancer/tumors (unspecified), gastric cancer, ovarian cancer, pediatric indications, sarcoma, and solid tumors. |
200 ,
21
| FRAGNAME | PDBID | SIMILIRITY | XSCORE | SMILE | HAC |
|---|---|---|---|---|---|
| 2woq_ligand_frag_4.mol2 | 2woq | 1 | -6.05 | CC(=O)O | 4 |
| 1r9l_ligand_frag_1.mol2 | 1r9l | 1 | -6.04 | CC(=O)O | 4 |
| 4euo_ligand_frag_2.mol2 | 4euo | 1 | -6.04 | CC(=O)O | 4 |
| 2nt7_ligand_frag_3.mol2 | 2nt7 | 1 | -6.02 | CC(=O)O | 4 |
| 2b4l_ligand_frag_1.mol2 | 2b4l | 1 | -6.00 | CC(=O)O | 4 |
| 1d6s_ligand_frag_1.mol2 | 1d6s | 1 | -5.99 | CC(=O)O | 4 |
| 1ibc_ligand_frag_15.mol2 | 1ibc | 1 | -5.99 | CC(=O)O | 4 |
| 1nms_ligand_frag_2.mol2 | 1nms | 1 | -5.99 | CC(=O)O | 4 |
| 2vl1_ligand_frag_2.mol2 | 2vl1 | 1 | -5.99 | CC(=O)O | 4 |
1717 ,
172

